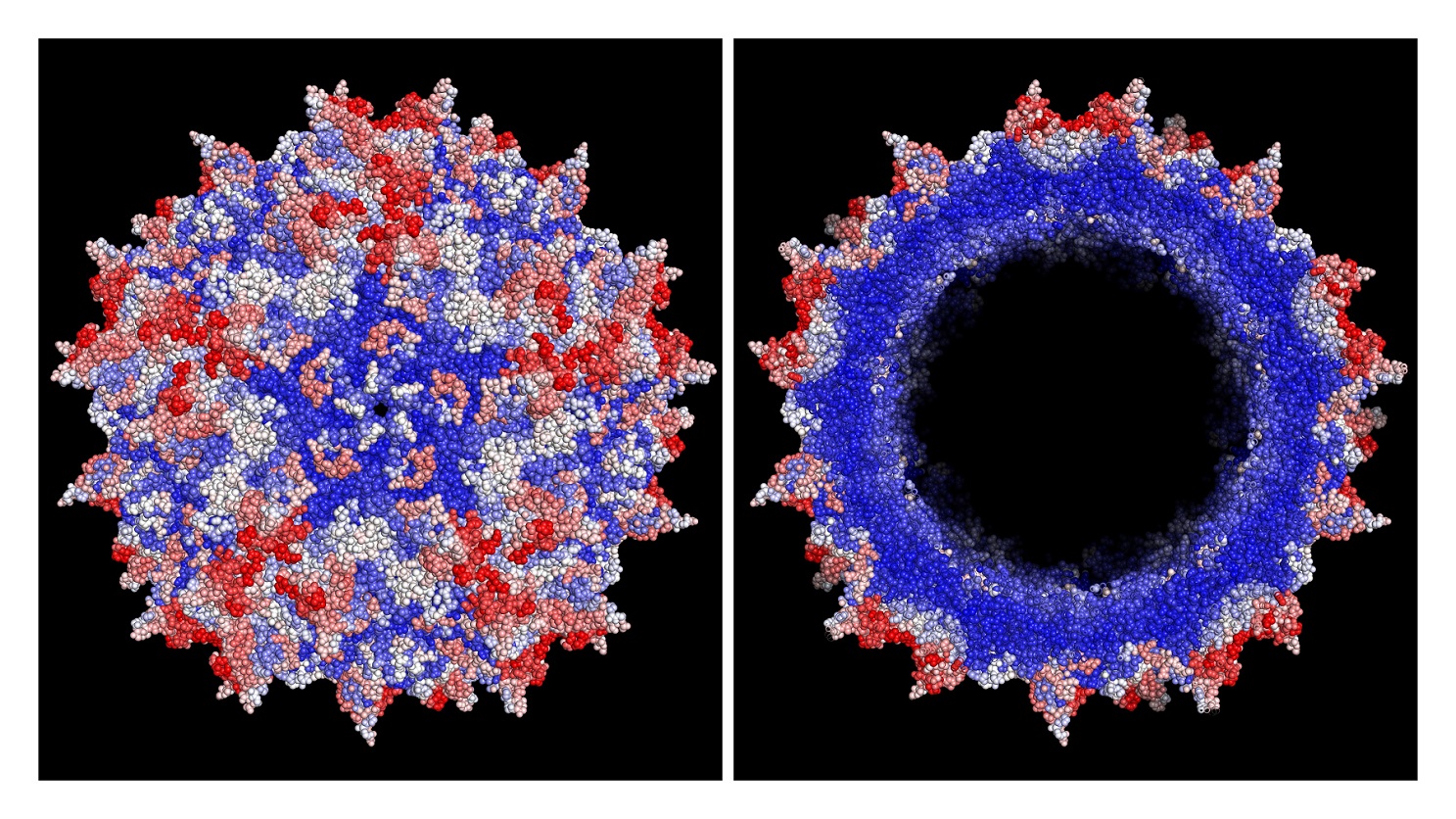
Improved AAV vector capsid for gene therapy engineered with a new machine-guided approach shows, in red, improvements in efficiency of viral production based on the average effect of insertions at all possible amino acid positions, with white showing neutral and blue showing deleterious positions. (Left: capsid viewed from outside, Right: cut-out to reveal inner positions). Credit: Eric Kelsic, Dyno Therapeutics
Dyno Therapeutics has raised $100 million, the third nine-figure financing announced by a gene therapy startup in the past seven days. CEO Eric Kelsic acknowledged the cash raised by Dyno’s peers, and he said it speaks to the importance of the problems that companies are trying to solve: delivery of gene therapies to more types of tissue so that these treatments can address more diseases and help more patients.
But as this field grows, Kelsic contends his firm has an advantage.
“Everyone wants to do what we do, but we got started earlier,” he said. “The key to machine learning is the more data you have, the better your models will be, and the more predictive.”
Gene therapies are delivered to their destinations in the body via an engineered virus. Adeno-associated virus (AAV) is the basis of the two FDA-approved gene therapies and it’s also the backbone of many gene therapies in development. This viral vector envelops the genetic cargo in a protein shell called a capsid.
But AAV as it’s found in nature isn’t optimal for therapeutic applications. It’s not suitable for delivery to certain types of tissue. Circulating throughout the body, it can trigger a dangerous immune response. Dyno and other startups are designing new capsids by employing artificial intelligence and machine-learning techniques to find the capsid properties that are best suited to therapeutic applications.
Dyno’s CapsidMap technology starts by designing millions of capsids and then running experiments on them, measuring for the properties that are relevant to a gene therapy, such as its ability to target a particular cell or its capacity for carrying a genetic payload. The technology then builds out a map of AAV capsids. Using computational tools, the company searches the map for capsids best suited for a particular therapeutic application.

A Deep-dive Into Specialty Pharma
A specialty drug is a class of prescription medications used to treat complex, chronic or rare medical conditions. Although this classification was originally intended to define the treatment of rare, also termed “orphan” diseases, affecting fewer than 200,000 people in the US, more recently, specialty drugs have emerged as the cornerstone of treatment for chronic and complex diseases such as cancer, autoimmune conditions, diabetes, hepatitis C, and HIV/AIDS.
CapsidMap stems from research at Harvard University, where Kelsic was a postdoc in the lab of geneticist George Church. Dyno formed in 2018 and received $9 million in seed funding from Polaris Partners and CRV. Inquiries from drug companies came soon after. Nearly a year ago, Dyno emerged from stealth and announced research partnerships with Novartis and Sarepta Therapeutics.
The deals call for Dyno to design the AAV vectors, which the companies will take into preclinical and clinical development. Novartis plans to use those vectors in gene therapies for eye diseases. Sarepta is interested in muscle disorders.
Specific financial details were not disclosed, but payments for research and milestones could bring Dyno more than $2 billion.
Last September, Roche signed on as a research partner, enlisting Dyno to design capsids for gene therapies that could address the central nervous system and the liver. The companies did not disclose specific financial terms for that deal except to say that Dyno could earn more than $1.8 billion in milestone payments.
Kelsic said the Series A financing will enable his company to expand CapsidMap so that it can design more capsids to address more tissue types. The next delivery targets are the heart, lungs, and kidneys. The Dyno team will also grow. Currently at about 50 employees, Kelsic projects headcount will triple in the next two years. As Dyno grows, Kelsic plans to add more research partners. But he added that the financing is not specifically to help the company find partners, it’s to fund the work to find new capsids that partners will want.
Other capsid-designing startups also have alliances with large pharmaceutical companies. Last week, Capsida came out of stealth with $50 million in Series A financing and a partnership with AbbVie that brings another $90 million. The Thousand Oaks, California-based startup is initially focusing on developing capsids that deliver gene therapies to cells of the central nervous system. On Monday, Affinia announced the close of a $110 million Series B financing. Affinia, which is based in Waltham, Massachusetts, is developing gene therapies for CNS and muscle disorders. It has a research partnership with Vertex Pharmaceuticals.
Dyno does not have a pipeline of its own gene therapies, and that is by design. The company has focused its efforts on developing its technology. Kelsic said that to expand the reach of gene therapy so that it helps more patients, the best way to do that is to build a technology platform that can attract a broad range of partners.
The Series A round of funding announced Thursday was led by Andreessen Horowitz. With the financing, Jorge Conde, general partner at that firm, is joining Dyno’s board of directors. Others participating in the financing include new investors Casdin Capital, GV, Obvious Ventures, and Lux Capital. Founding investors Polaris Partners, CRV, and KdT Ventures also participated in the round.












