
Although it’s only been a little more than one month since President Donald Trump took office, it feels like it’s been longer than that. Much remains to be seen with how the administration will handle healthcare. But what’s in store for the medical device industry and what can we expect in the way of changes to the regulatory landscape?
Here are a few predictions and observations to think about as we near the end of the first quarter of 2017.

The Power Behind Enterprise EHR Software for Large Healthcare Systems
Enterprise EHR boosts scalability, interoperability, and governance for large healthcare systems.
A new FDA Commissioner in 2017
A new president often results in a new FDA commissioner. This election cycle was no different. Dr. Robert Califf resigned his post after Donald Trump took office.
Again, this is not all that unusual when a new president takes office.
There are currently a couple of names floating around as possible candidates for the new FDA commissioner post. Interestingly, the two names floating around are both involved with venture capital investment.
Prediction: Expect the new FDA Commissioner to have a venture capital background.
One of the names rumored to fill the post is Jim O’Neill. He is currently a managing director with Mithril Capital Management. Interestingly, O’Neill is not a medical doctor, and while not technically a pre-requisite for FDA commissioner, it is often the case.
The possibility of O’Neill as FDA Commissioner is interesting and likely would have a large influence on pharma regulation. In fact, O’Neill has stated that drugs should be allowed on the market before their efficacy has been established, as long as they are proven to be safe.
The other name currently being thrown around is Dr. Scott Gottlieb. Gottlieb is currently a venture capitalist with New Enterprise Associates. Gottlieb seems like he would be a better fit than O’Neill, considering his M.D. background and prior experience as deputy commissioner of the FDA.
At the time Califf resigned, he had only been on the job since February 2016, so his imprint on the FDA was too new to have much of a lasting impression. Califf had been identified as a “big data” advocate. It’s not clear if either O’Neill or Gottlieb will endorse that position. Regardless, I suspect that either O’Neill or Gottlieb (or another unnamed candidate) will be feeling pressure to get new products — drugs and devices — cleared more quickly.
Verdict: I predict that a new FDA commissioner will bode well for the medical device industry.
CAPA (corrective action/preventive action) is a continued area of concern
CAPA is a major focus during FDA inspections as well as ISO audits. This has been the case for quite some time.
Why?
Many view the CAPA process as the most critical quality management process for medical device companies. CAPA is the process within a company that provides oversight and visibility into all the ways to improve and fix issues.
Prediction: CAPA will continue to be a hot topic during FDA inspections and ISO audits in 2017.
How a company embraces CAPA can be a make or break scenario. In some cases, companies overuse CAPA. They realize how important the CAPA process is in the grand scheme of things — so much so that CAPA is used to capture many things. Maybe too many things.
Other companies do not leverage CAPA enough. For these medical device companies, CAPA is sometimes an afterthought.
In both situations, CAPA is not being used as intended. CAPA is intended to be a gateway to identifying opportunities for improvement, opportunities to prevent issues, and/or ways to correct problems.
Know that CAPA will be a key part of any FDA inspection and ISO audit. Here is a link to data from FDA’s FY2016 regarding 483 observations. Note the number of citations logged under FDA 21 CFR 820.100, the regulatory code corresponding to CAPA.
ISO 13485:2016 will become mainstream
Looking back on 2016, it was a big year for ISO 13485 — the medical device quality management standard. A revision to this standard was long overdue and published in March 2016.
There is a three-year grace period to transition from old ISO 13485:2003 to the new ISO 13485:2016. But you should expect ISO 13485:2016 to begin to take hold in 2017.
Prediction: ISO registrars will begin transitioning certifications to ISO 13485:2016 in 2017.
In fact, I know of several ISO audits being scheduled for companies in early 2017 where the registrar is requesting that the 2016 “upgrade” take place.
Beware. Do not just assume that your quality management system (QMS) is already in alignment with ISO 13485:2016. This new revision to the standard does include several changes.
I recommend that you conduct a gap analysis of your current QMS against ISO 13485:2016 and define a quality plan for making this transition.
EU medical device regulations and IVD regulations finalized
The European Commission has long discussed changes that impact the medical device industry. Currently in the EU, medical devices and in vitro diagnostics (IVDs) are governed according to directives (MDD and IVDD).
Prediction: EU will implement medical device regulations and IVD regulations (IVDRs) in the second quarter of 2017.
The European Council released the final draft of its long-awaited Medical Device Regulations (MDR) and In Vitro Diagnostics Regulations (IVDR) in late February, with voting on them set to begin in March.
Yes, these new EU regulations will have an impact on medical devices and IVDs sold in the European market. Yes, there is a transition period expected for both new regulations. MDRs go into full effect within 3 years and IVDRs within five years of adoption.
While this seems like you have plenty of time to adjust, I encourage you to stay on top of these changes. These changes will not only impact new products but also any existing products which are already CE marked.
Expect FDA 510(k) review times to increase
In the U.S., the most common pathway to receive FDA market clearance for a medical device is a 510(k) submission.
The 510(k) process is well-established and defined. In recent years, the FDA instituted new policy for 510(k)’s known as the “refuse to accept” policy.
As a result of implementing this policy, 69 percent of 510(k)s were rejected during the first half of 2015.
Prediction: FDA 510(k) review times will continue to increase in 2017.
While the final 2016 data is still not available, I would like to share some trends from 2015.
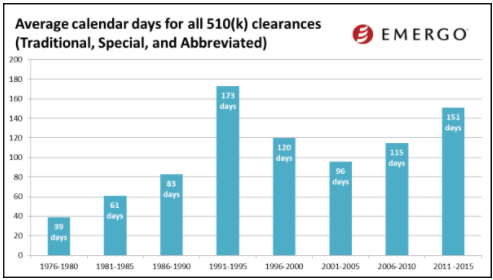
(image courtesy of Emergo Group)
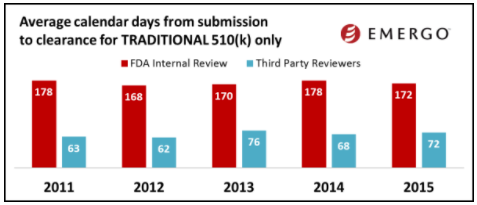
(image courtesy of Emergo Group)
The charts above show how 510(k) review times have been trending over the past several years. Based on this data, you might conclude that 510(k) review times have been fairly consistent over the past few years. Keep in mind this data does not factor in those 510(k) applications that are rejected. When this happens, the clock starts over.
Photo: KatarzynaBialasiewicz, Getty Images,
Jon Speer is the founder and VP of QA/RA at Greenlight Guru, a software company that produces the only modern quality management software solution exclusively for medical device companies. Device makers in hundreds of cities in more than 30 countries use Greenlight Guru to get safer products to market faster while pushing beyond compliance to True Quality.
Jon is a medical device industry veteran with over 20 years experience having helped dozens of devices get to market over his career in a variety of roles including product development, project management, quality and regulatory. He is a thought leader, speaker and regular contributor at numerous leading industry publications. He is also the host of the #1 most downloaded podcast in the industry, The Global Medical Device Podcast.
This post appears through the MedCity Influencers program. Anyone can publish their perspective on business and innovation in healthcare on MedCity News through MedCity Influencers. Click here to find out how.

