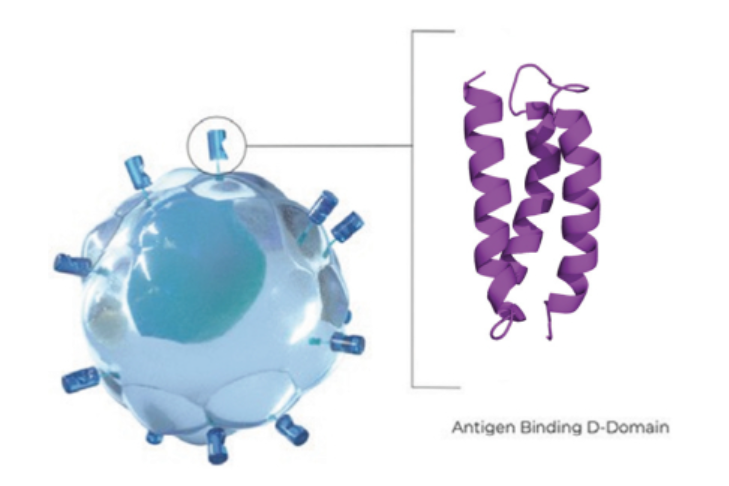
Gilead and Roche Bet on Protein Degraders for Their Cancer Drug Pipelines
Gilead Sciences exercised its option to license a Kymera Therapeutics’ protein-degrading drug designed to eliminate the cancer-driving protein CDK2. Meanwhile, Roche’s new partnership with C4 Therapeutics is focused on developing degrader antibody drug conjugates for undisclosed cancer targets.