Intellia’s Data Reveal Tees Up FDA Filing for CRISPR-Based In Vivo Gene-Editing Med
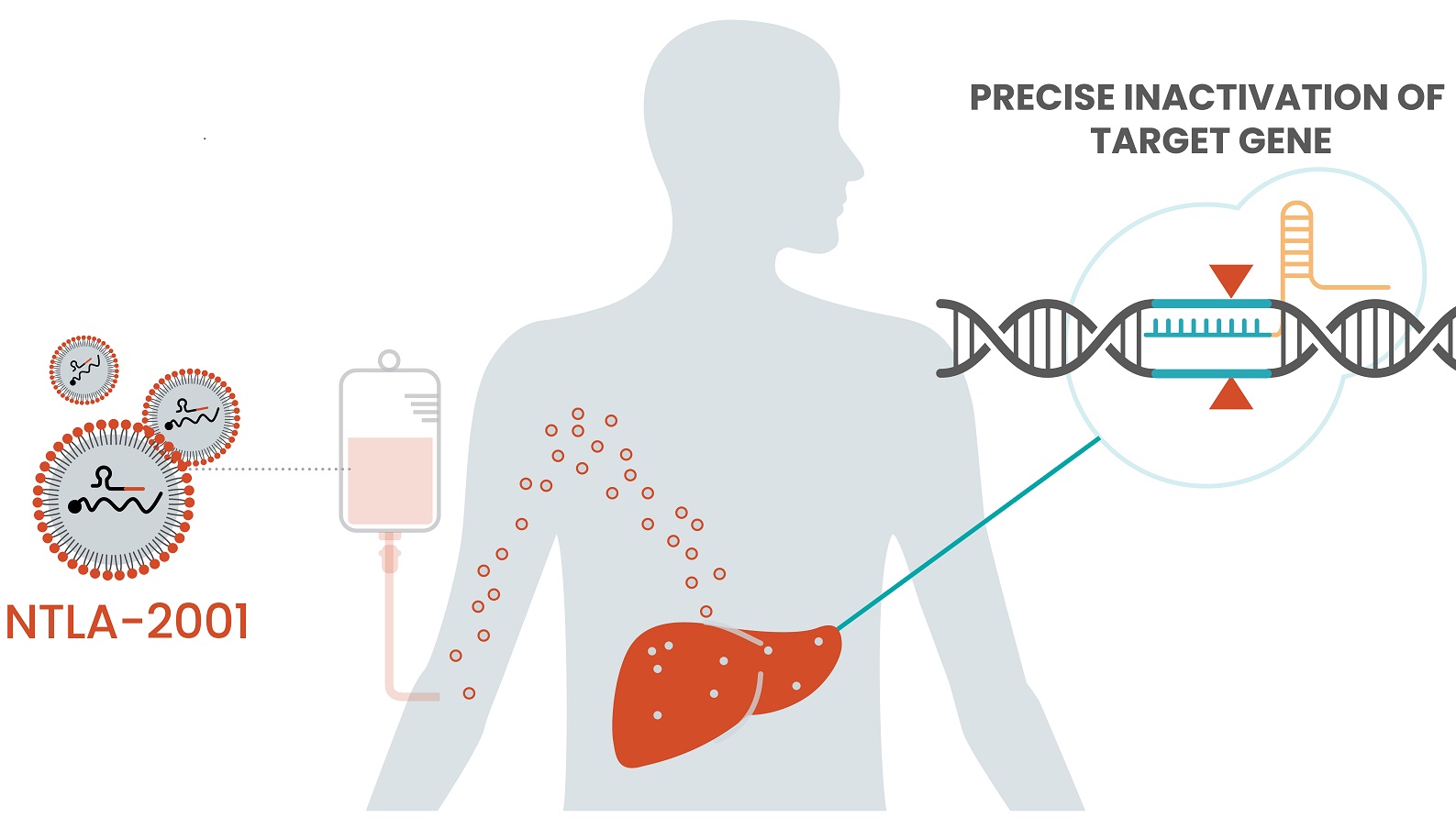
Intellia Therapeutics’ lonvo-z uses CRISPR to inactivate a gene in the liver to reduce levels of a protein key to the swelling attacks from the rare disease hereditary angioedema. Intellia has filed a regulatory submission for this genetic medicine, which could become the first in vivo gene-editing therapy to land FDA approval.