
CEO of Otsuka North America: Are We Getting better access with AI?
In an interview, Rabah talked about the promise of AI, where it can have the most impact, while also responding to the IRA and high drug prices in the United States.

In an interview, Rabah talked about the promise of AI, where it can have the most impact, while also responding to the IRA and high drug prices in the United States.

Otsuka Pharmaceutical is acquiring Jnana Therapeutics, whose lead drug is a potential treatment for the rare metabolic disorder phenylketonuria (PKU). Jnana’s drug could provide an alternative to two FDA-approved PKU medicines from BioMarin Pharmaceutical.

Small practices play a critical role in healthcare delivery, but they cannot continue to absorb ever-increasing administrative demands without consequences.
Rexulti, a drug for schizophrenia and depression, now has an additional approval for treating agitation in Alzheimer’s disease patients. The FDA decision makes the Otsuka Pharmaceutical and Lundbeck product the first drug approved for this indication.

Otsuka Pharmaceutical is ending its alliance with Akebia Therapeutics, a move that comes less than two months after the FDA rejected their partnered anemia drug, vadadustat, and asked for another clinical trial. Otsuka alleges unspecified breaches of the agreement by Akebia, and ending the alliance early could spare it from spending hundreds of millions of dollars more on the drug.

The companies said Monday that they would partner to develop small-molecule oncology drugs against several targets, including KRAS mutations, long thought "undruggable." Amgen and Mirati have already presented clinical data from their KRAS programs.

Several FDA approvals - including ones going against AdCom votes - have attracted controversy and a perception of lowered standards at the agency. However, some see a long-standing institutional tension between patient protection and speedy approval, with the latter having a net positive effect.

Veradigm examines key clinical trends, comorbidity profiles, and treatment trends across adolescence, reproductive years, and peri-/post-menopause. Download it today!

In an article published in the journal mAbs Monday, executives from Adimab alleged that MIT's Ram Sasisekharan copied prior monoclonal antibody research. Visterra, a company Sasisekharan founded that had been developing one of the antibodies, was acquired by Otsuka for $430M last year.

A new report by Accenture suggests that over the next three years, 54 percent of biopharma industry revenues could come from new advancements in medical science and technology - and combinations of the two.

Andrew Wright, Otsuka America Pharmaceutical Vice President, digital medicine and medical device division, and David O'Reilly Proteus Digital Health Chief Platform Officer, shared some insights on what it takes for a collaboration to succeed, especially when you are doing something that's never been done before.
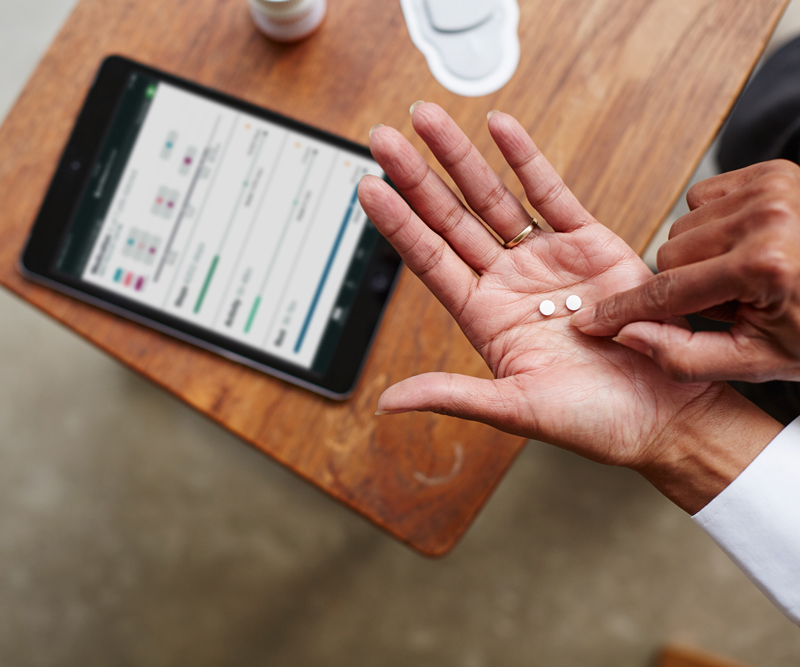
The FDA's approval of Abilify Mycite, which is expected to come to market early next year marks a milestone for mental health patients but also raises some interesting questions about how the healthcare industry will respond to the digital pill tech.

Abilify MyCite, developed in collaboration between Proteus Digital Health and Otsuka Pharmaceutical, has an ingestible sensor embedded in the pill which records that the medication was taken. It reflects a trend by companies to use sensors to quantify the effectiveness of medications.