
FDA clears Propeller Health sensor for GSK Ellipta inhaler
Propeller and GlaxoSmithKline announced a joint development agreement less than a year ago.

Propeller and GlaxoSmithKline announced a joint development agreement less than a year ago.

The FDA has more than 700 job vacancies in its division that reviews new drugs, and the agency has been under fire for taking too long to approve medications.

Munck Wilson Mandala Partner Greg Howison shared his perspective on some of the legal ramifications around AI, IP, connected devices and the data they generate, in response to emailed questions.

TytoCare will bundle the stethoscope with other FDA-listed medical devices for healthcare professionals or even consumers to initiate telehealth exams.

Oregon Health & Science University researchers published their findings Tuesday in The BMJ.

Critics say getting generic alternatives to the U.S. market for products like EpiPen is still taking far too long.

Theranos confirmed the withdrawal to the Wall Street Journal, but didn't say anything about the FDA inspection. Instead, company officials tried once again to put on a happy face.
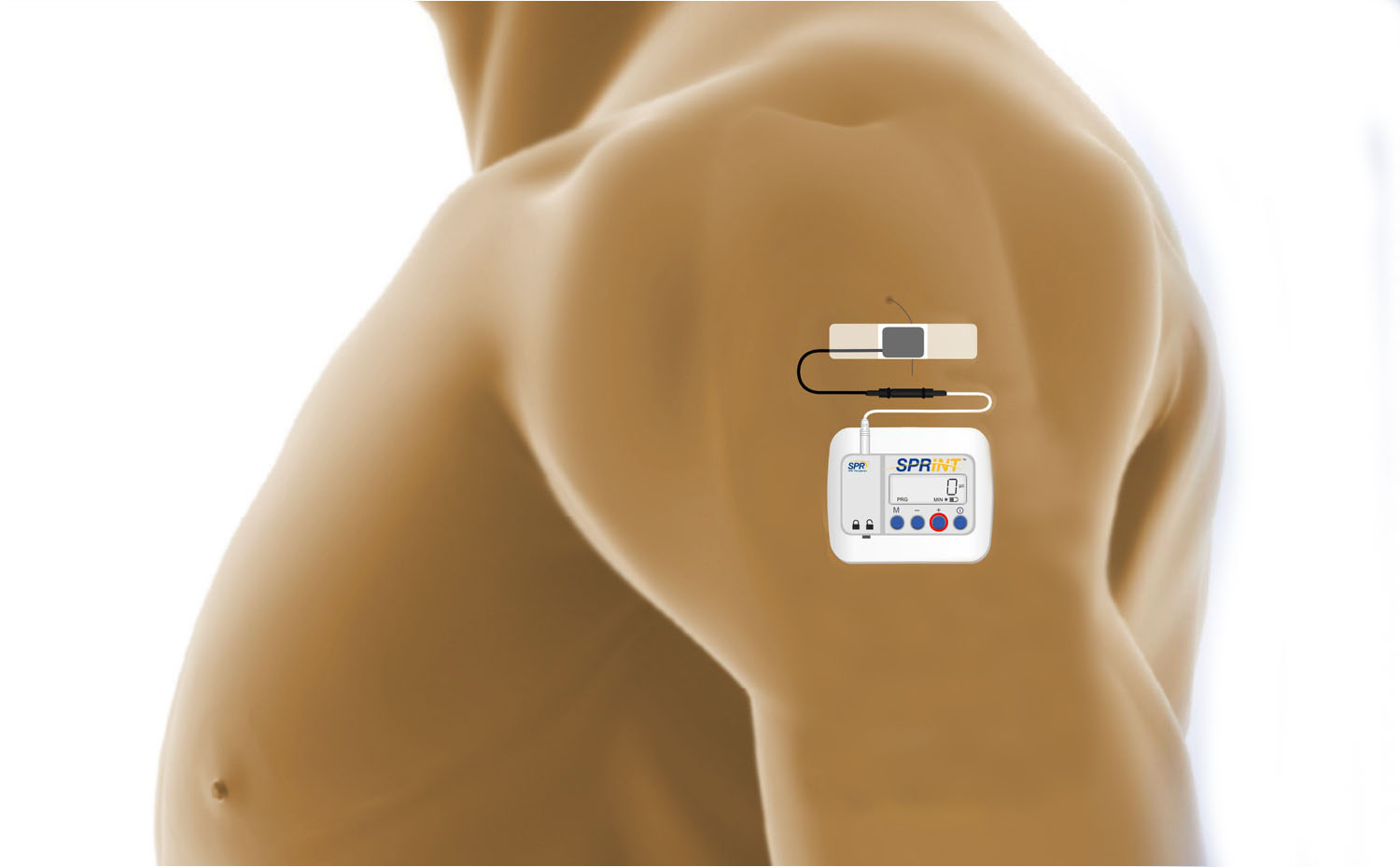
SPR Therapeutics has developed a peripheral nerve stimulation system that is placed percutaneously through the skin instead of being implanted and which can be completely removed from the body after therapy period.

The company’s internal emails, filed in a Pennsylvania court this month as part of a patient lawsuit, reveal conflicts inside Olympus over how to respond to a growing threat to patient safety, pitting U.S. executives against their superiors in Japan who had the final say.
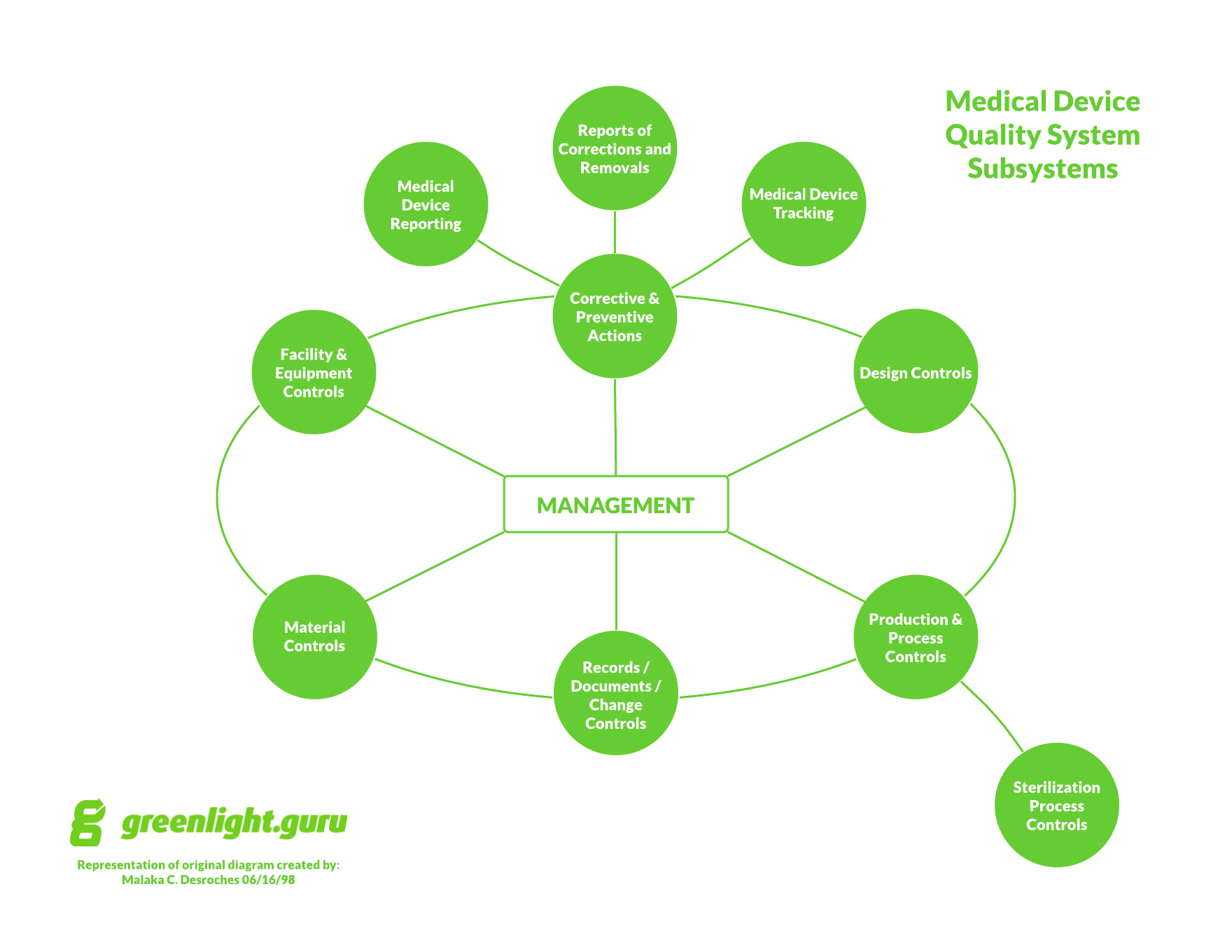
Medical device companies, listen up. There is zero excuse for not complying with medical device quality system regulations. Zero. Why? FDA has published and makes available ALL regulations required for medical device companies. Look them up by searching 21 CFR part 820. And for outside U.S., you can easily obtain ISO 13485 for a relatively […]
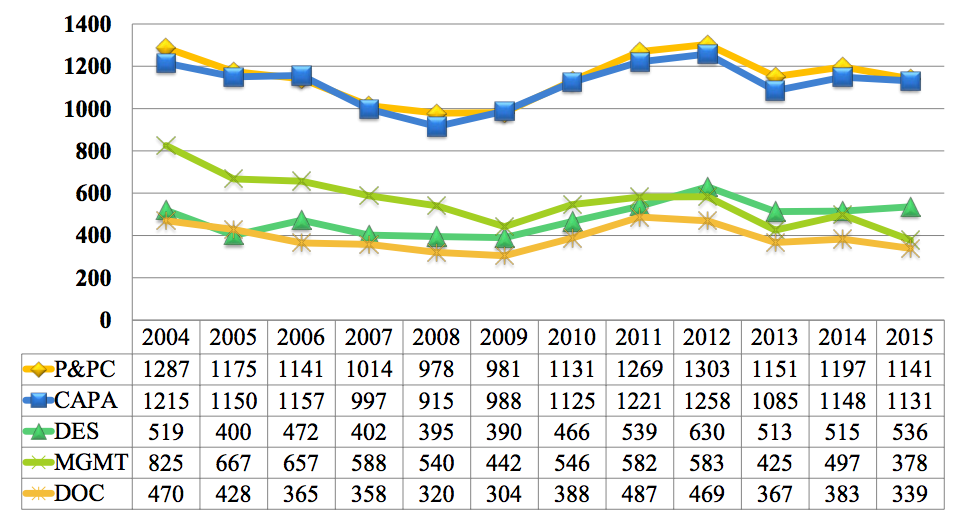
When was your last FDA inspection? You may believe that the likelihood of an FDA inspection happening to your company is low. However, medical device companies registered with FDA with class II and class III products are subject to mandatory inspections at least once in every two year period. FDA Medical Device QS Surveillance Inspections […]

We will highlight Build My Health's revenue practice management tools, which could help physician practices add up to $250,000 to their practices.
Also, the FDA has approved an investigational test to screen blood for the Zika virus and Consumer Reports explores the mystery of bad doctors.

Plus, biopharma CEOs grow bearish about their ability to raise new capital, Hollywood Presbyterian ransomware attack may be just the beginning and incoming Dana-Farber CEO is excited for the future.

Also, Sean Parker donates $10 million for diabetes, a look at the FDA's lab-testing regulation plans, the potential for a notable New England healthcare merger, and a CES preview (really? A CES preview in November).

The Food and Drug Administration on Friday issued an alert warning that Hospira's Symbiq Infusion System smart pumps could be vulnerable to hackers,a and recommended that healthcare facilities phase out use of the devices.

There were more than 4,000 observations for medical device companies during FDA inspection activities in 2013.