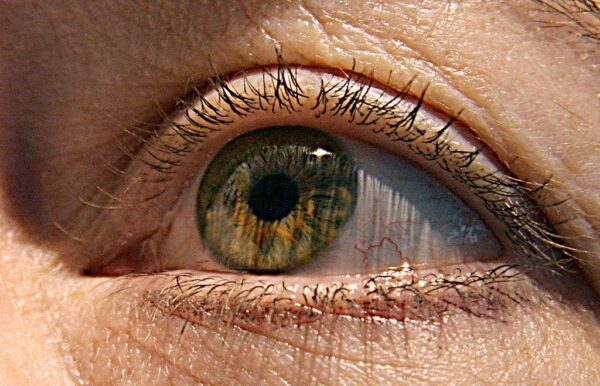
Bausch + Lomb is expanding its ophthalmology portfolio and pipeline, paying Novartis $1.75 billion to acquire a commercialized drug for dry eye disease along with two experimental eye products.
The sum announced Friday is an upfront payment. Another $750 million is tied to the achievement of milestones. The main asset of the deal is Xiidra, an eye drop that alleviates the inflammation associated with dry eye disease. Per deal terms, Bausch + Lomb will take on the Novartis salesforce that supports Xiidra.

The Hidden Administrative Tasks Draining Small Practices
Small practices play a critical role in healthcare delivery, but they cannot continue to absorb ever-increasing administrative demands without consequences.
Bausch + Lomb already has a presence in dry eye disease following FDA approval in May of its drug, Meibo. That eye drop’s key ingredient is a compound that forms a layer on the tear film, preventing the evaporation that contributes to dry eye disease. Bausch + Lomb says Xiidra and Meibo will complement each other, targeting different parts of the dry eye disease cycle. Xiidra is small molecule that binds to LFA-1, a protein on the surface of immune cells called leukocytes. Doing so prevents LFA-1 from interacting with another protein that contributes to an inflammatory response in the eye.
Xiidra was originally developed by Shire, which won FDA approval for the drug in 2016. After Takeda Pharmaceutical acquired Shire in 2019, the Japanese drug giant sold off some of Shire’s assets. Novartis bought Xiidra from Takeda for $3.4 billion up front in 2019. Now Novartis is offloading the product.
Xiidra generated $487 million in revenue in 2022, a 4% increase from the prior year, according to Novartis financial reports. But those sales are well short of the blockbuster potential that Novartis touted when it acquired the eye drug. An approval in Europe that could further boost sales did not happen. In 2020, Novartis withdrew an application seeking regulatory approval there due to questions from regulators.
Bausch + Lomb’s deal with Novartis includes libvatrep (formerly SAF312), an experimental treatment for chronic ocular surface pain. This small molecule drug is designed to block TRPV1, a receptor associated with pain. Results from a Phase 2b test are expected in the third quarter of this year. The other pipeline product coming to Bausch + Lomb is AcuStream, a medical device being developed for precise dosing and accurate delivery of topical eye medications.
“This acquisition is a prime example of our strategy in action, as it provides needed scale for the company and transforms our pharmaceuticals business by making us a leader in ocular surface diseases,” Bausch + Lomb Chairman and CEO Brent Saunders said in a prepared statement.
Bausch + Lomb is paying for the Novartis eye products with debt financing from J.P. Morgan. The acquisition is expected to close by the end of this year.
Photo: Karen Bleier/AFP, via Getty Images








