Medical device maker Vertos Medical raked in $26 million on Wednesday in a Series C funding round led by Norwest Venture Partners. The company, which is based in Aliso Viejo, California, develops minimally invasive devices to guide procedures for lumbar spinal stenosis (LSS) — a condition characterized by the narrowing of the spinal canal in the lower back.
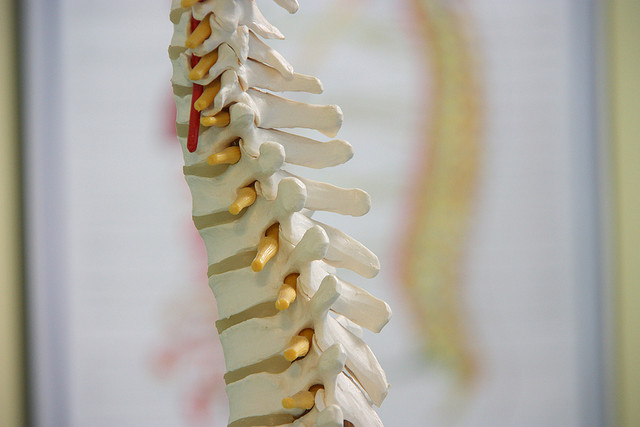
LSS is commonly associated with age-related degenerative changes in the spine, such as the growth of bone spurs or thickening of ligaments. The narrowing of the spinal canal can lead to compression of the spinal cord and nerves, causing back and leg pain, as well as numbness and weakness. Treatment options range from conservative measures such as physical therapy and pain medication to surgical interventions in severe cases.
Through its products, Vertos’ is seeking to help patients suffering from LSS-induced back pain reclaim their quality of life, CEO Eric Wichems said in an interview.
“Allowing patients to get back to what matters most to them is what matters most to us. From playing with their grandkids or golfing with friends to getting a better night’s sleep and doing it all over again, we want to help patients find joy every day by increasing their mobility and reducing their pain — in the least invasive way,” he declared.
The company’s flagship product is called “mild” — it’s a minimally invasive procedure that removes small pieces of bone and ligament. The image-guided procedure is performed in an outpatient setting with no general anesthesia required.
“The purpose of the procedure is to remove small amounts of bone and ligament that are pushing into the spinal canal and pinching the spinal cord. Some have described this squeezing phenomenon as a kink in a drinking straw. This pinching causes pain and numbness in your back and legs while you are standing or walking,” Wichems explained.

The Hidden Administrative Tasks Draining Small Practices
Small practices play a critical role in healthcare delivery, but they cannot continue to absorb ever-increasing administrative demands without consequences.
During the mild procedure, the physician inserts a hollow metal tube about the size of a straw in a patient’s back, and X-ray guidance leads them to the area where the ligament or bone is causing the problem. The physician then inserts specialized instruments through this tube to remove small amounts of ligament and bone. Once this is done, the physician removes the instruments and the small tube.
The procedure leaves no implants behind, nor does it require stitches. Patients go home the same day and can typically resume activities of daily living within 24 hours, Wichems said.
He noted that more than 1,000 physicians have performed at least one mild procedure during the first quarter of this year. Mild is a single-use device system, so doctors need to buy a device for each patient that receives the procedure.
Vertos’ target physician is one who has experience doing procedures using X-ray guidance. This is usually an interventionist, such as an interventional pain physician, interventional radiologist or a physiatrist, Wichems pointed out.
The company’s target patient is someone who did not benefit from conservative care options and is looking for a procedure that removes the root cause of the stenosis. Typically, these are patients who are not candidates for more invasive surgeries or choose not to get the surgery, Wichems explained.
In his view, epidural steroid injections are Vertos’ biggest competitor. The mild procedure has a similar safety profile to these injections — however, the procedure removes the cause of the problem, whereas the injections simply alleviate the pain, Wichems pointed out.
Some patients with LSS get epidural steroid injections in a cyclical nature — they go back to get one each time their pain resurfaces. One of Vertos’ biggest challenges will be getting these patients “to consider doing a procedure that treats the underlying condition rather than going home and accepting their limited mobility and associated diminishing quality of life,” Wichems said.
Photo: Michael Dorausch