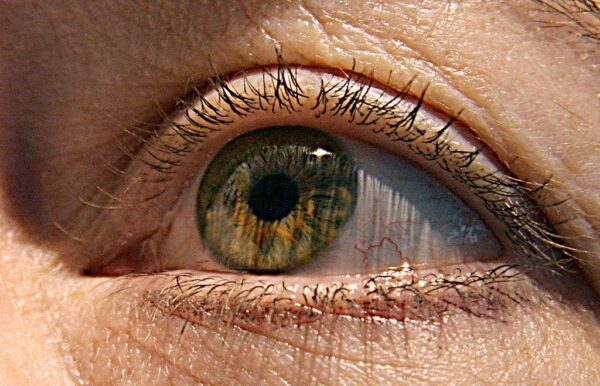
An experimental MeiraGTx gene therapy that has clinical data showing reversal of blindness in children born with a rare, inherited eye disorder is heading to Eli Lilly.
According to the terms of the agreement announcement Monday, Lilly is paying $75 million up front for global rights to the gene therapy, AAV-AIPL1. MeiraGTx could receive up to an additional $135 million in near-term payments tied to regulatory and approval milestones. The New York-based biotech is currently readying this gene therapy for submissions seeking regulatory approvals in the U.S. and Europe.
AAV-AIPL1 was developed to treat Leber congenital amaurosis 4 (LCA4) caused by genetic mutations in the AIPL1 gene. AIPL1 protein is important for proper function of the rod and cone photoreceptors of the eye. Mutated versions of this protein lead to retinal degeneration. LCA4 patients either have severe vision impairment at birth or develop it shortly afterward. There are currently no treatments for LCA4.

The New Blueprint: How Clever Care Health Plan is Scaling Its Member Experience [Video]
MedCity News was at the Vive conference and spoke with executives who shared their insights for the healthcare industry.
The MeiraGTx gene therapy uses an adeno-associated viral vector to deliver functional copies of the AIPL1 gene to the photoreceptors of the central retina. The one-time treatment is administered by subretinal injection. MeiraGTx tested its LCA4 gene therapy in an open-label, single-arm study that enrolled 11 children between the ages of 1 and 4. All participants were legally blind at birth; their only visual acuity was the perception of light.
This past February, MeiraGTx reported clinical trial results showing all participants experienced gains in visual acuity in the treated eyes four or more weeks after receiving the gene therapy. In addition, patients showed improvement in functional vision and preservation of the retinal structure compared to the untreated eye. No adverse effects were reported. Detailed results were published in the journal The Lancet.
Under the agreement, MeiraGTx could receive up to $400 million in milestone payments from Lilly, a sum that includes the up to $135 million in near-term payments. If the gene therapy is approved, the biotech would also receive royalties from Lilly’s sales of the commercialized product. The deal for AAV-AIPL1 starts a broader collaboration between the two companies. According to a MeiraGTx regulatory filing, Lilly also gains rights to develop and commercialize preclinical therapeutic candidates that MeiraGTx has in development for other inherited retinal dystrophies. Those diseases were not disclosed.
The filing states Lilly has an exclusive license to proprietary MeiraGTx intravitreal capsids (the protein shells that enclose the genetic cargo of a gene therapy and target its delivery to particular tissues in the body) for use with up to five ophthalmologic targets selected by Lilly. The pharma company also gets an exclusive license to proprietary pan-retinal or rod-specific promoters for use with up to five targets to be selected by Lilly. MeiraGTx is also granting Lilly certain rights to use its proprietary riboswitch technology for gene-editing applications in the eye. Riboswitch enables in-vivo production of a therapeutic protein or gene-editing nuclease. This production is controlled by an oral small molecule.

The Hidden Administrative Tasks Draining Small Practices
Small practices play a critical role in healthcare delivery, but they cannot continue to absorb ever-increasing administrative demands without consequences.
Gene therapy is a growing part of Lilly’s pipeline. In 2021, the pharma giant acquired Prevail Therapeutics, developer of gene therapies for neurodegenerative and rare disorders. Prevail has since added a multi-drug R&D alliance with startup Capsida Biotherapeutics spanning gene therapies for central nervous system (CNS) disorders. In 2022, Lilly bought Akouos, developer of a gene therapy for a rare, inherited form of hearing loss. Earlier this year, Lilly struck a deal to secure rights to a Sangamo Therapeutics capsid for delivery of genomic medicines addressing CNS disorders. And last month, Lilly acquired Adverum Biotechnologies, whose lead program is a gene therapy in Phase 3 testing for the wet form of age-related macular degeneration.
In a prepared statement, Andrew Adams, Lilly group vice president, molecule discovery, said ophthalmology is an emerging area of interest for his company.
“We are excited to partner with MeiraGTx to bring transformative treatments to patients around the world suffering from eye diseases, starting with AAV-AIPL1, which has shown the unprecedented ability to restore vision in children who were born legally blind,” he said.
Photo: Karen Bleier /AFP, via Getty Images






