
What will digital health M&A look like in 2022?
After a busy year for digital health, four experts shared their predictions on which sectors will see the most M&A, and which companies are likely to get together in the future.
Insurers that didn’t meet medical loss ratio requirements have paid out a total of $2 billion in rebates to nearly 10 million people, split between people who received their insurance through individual market plans and group plans.

The company would become part of GE’s ultrasound business, expanding its capabilities to include surgical visualization. GE plans to spin out its healthcare division into a publicly traded company in 2023.

Digital health companies have now raised more than $20 billion in 2021, more than any other year. While experts expect to see the dealmaking continue, they raised caution about the challenges of going public with high valuations.

Oracle is reportedly in talks to buy Cerner for $30 billion, according to the Wall Street Journal. If it closes, it would be the largest health tech deal this year.

The insurers referred to deals Gilead struck with Janssen, Bristol Myers Squibb to prevent the generic version of its retroviral medications from being used as part of combination treatments. They also allege the company struck a deal with Teva to delay the release of its generic drug.
The company received a warning letter on Wednesday related to inadequate quality requirements at the headquarters for its diabetes business. It comes after Medtronic expanded a recall of its MiniMed 600 series insulin pumps.

The startup is looking to help launch health plans by handling some of the administrative components. It bills itself as a tech-forward third-party administrator, and can help manage claims and enrollment.

The company, which is building a cloud-based platform to help health systems pull health records together, recently closed a $150 million funding round.

A specialty drug is a class of prescription medications used to treat complex, chronic or rare medical conditions. Although this classification was originally intended to define the treatment of rare, also termed “orphan” diseases, affecting fewer than 200,000 people in the US, more recently, specialty drugs have emerged as the cornerstone of treatment for chronic and complex diseases such as cancer, autoimmune conditions, diabetes, hepatitis C, and HIV/AIDS.
A panel of judges on the 8th Circuit Court of Appeals declined to reinstate a vaccine mandate for healthcare workers that was set to go into effect in January. A federal judge had blocked the mandate last month from the Centers for Medicare and Medicaid Services after 10 states filed suit.

The American Medical Association, American Hospital Association and other provider groups are suing the Department of Health and Human Services over a small but important detail of how it is implementing surprise billing legislation. They argue that the arbitration process for unresolved disputes currently favors insurers.

According to Rock Health’s annual consumer adoption survey, telehealth users tend to be higher income, younger and live in urban areas. The survey also tracked people’s satisfaction with telehealth and what modalities people used most to access care.
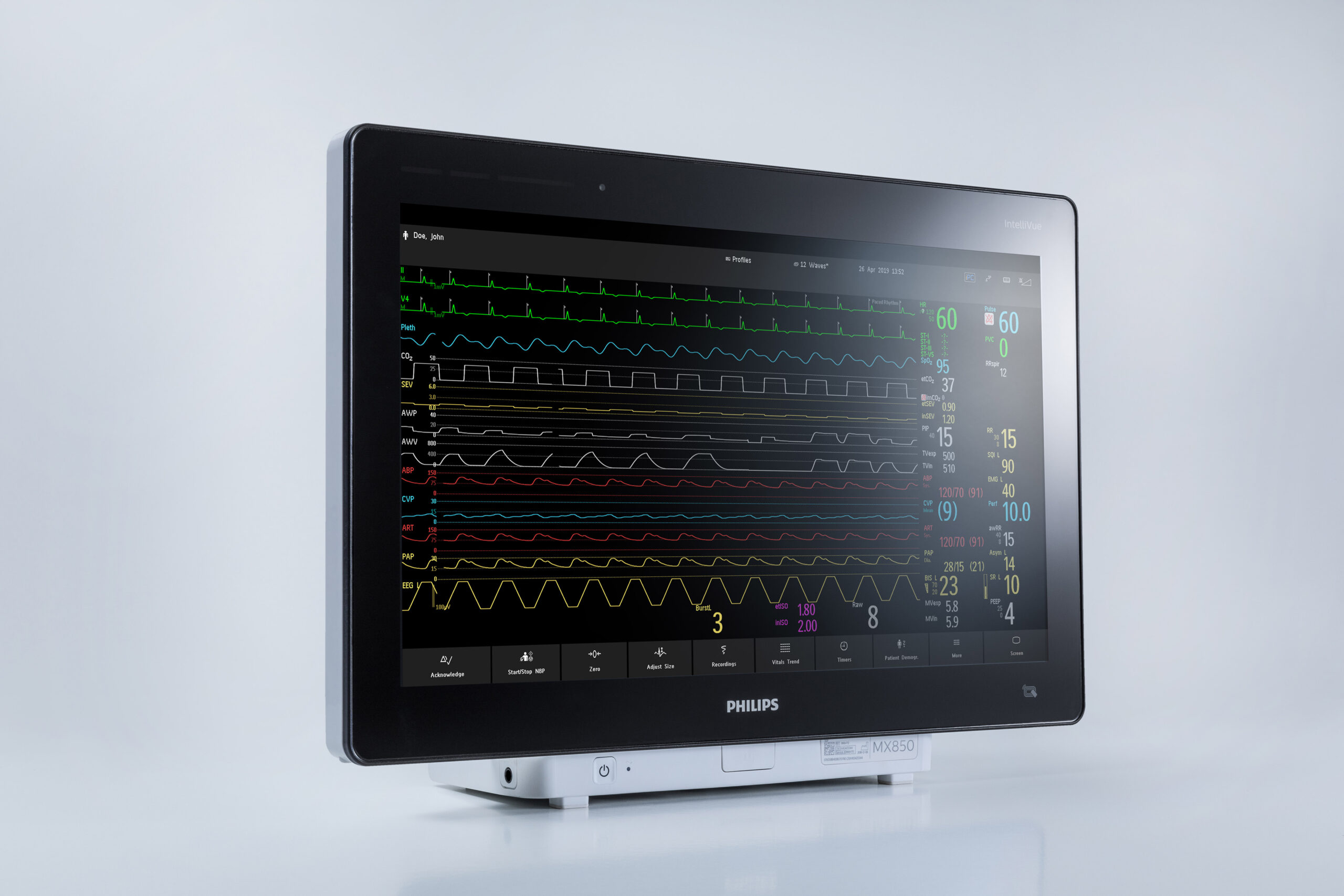
The FDA granted 510(k) clearance to two of Philips’ newest acute care patients monitors, the IntelliVue MX750 and MX850. A year ago, the company had gotten an emergency use authorization for its monitors.

Cigna Ventures and Bright's majority shareholder, New Enterprise Associates, are contributing to the financing. It comes as Bright faces higher-than-expected medical costs during the pandemic.