
MacroGenics’ stock price more than doubles on successful breast cancer study
In a conference call with analysts, the company's CEO was tight-lipped about the median improvement in PFS, but the results nevertheless beat many investors' expectations.

In a conference call with analysts, the company's CEO was tight-lipped about the median improvement in PFS, but the results nevertheless beat many investors' expectations.

The agency plans to release several guidances on complex generics and biosimilars, along with taking a deep dive into the Orange Book patent and exclusivity database.

Munck Wilson Mandala Partner Greg Howison shared his perspective on some of the legal ramifications around AI, IP, connected devices and the data they generate, in response to emailed questions.

Generics industry trade group the Association for Accessible Medicines anticipates that the CREATES Act could see action in Congress next year amid bipartisan support for tackling drug costs.
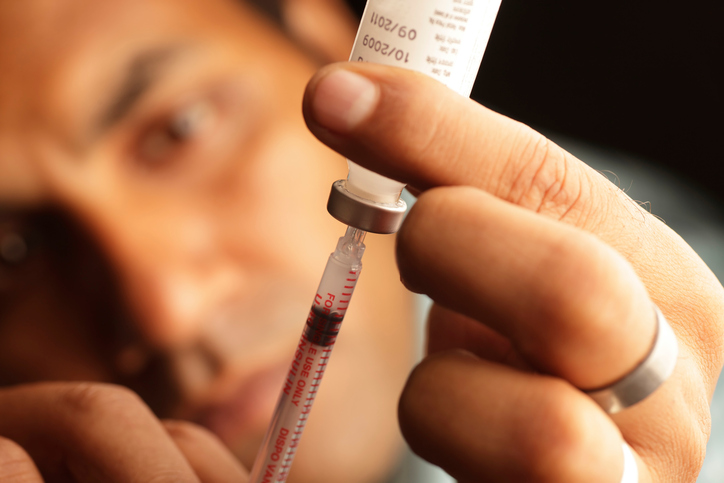
Insulin prices have shot up nearly 600 percent, according to a congressional study, leading to potentially deadly rationing by many patients.

The FDA last week issued the draft guidance, under which it would publicly state if it denied branded drugmakers' citizen petitions because they were designed to delay generic market entry.

Expert says that biosimilars' pricing discounts and lower-than-expected patient populations could make the market less attractive for smaller players.

The study can assure clinicians that Sandoz's Zarxio works just as well in febrile neutropenia as originator Amgen's Neupogen, author says in interview.

Soliris, the drugmaker's cash cow approved for the same disease, faces biosimilar competition as patent expiry looms.

Most drugs named are biologics, and manufacturing trade secrets make competition especially tricky, legal expert says.

Agency will create new review tools and other resources to encourage development of biosimilars.

We will highlight Build My Health's revenue practice management tools, which could help physician practices add up to $250,000 to their practices.

Market forecast rosy, though market in United States remains young.

Here's what is preventing biosimilars, which are growing in importance, from being more widely available in the U.S.

Oncobiologics has no home ground advantage. There is no clearly defined path with the FDA, EMA or any other regulatory agency. And it’s battling against the largest biotech and pharma companies in the world as they work to extend their patent exclusivity. How can it survive and bring biosimilars to the market at 50% of the cost?

Drug patents are always more complicated than they seem, but a new infographic from Dickson Data spells out some of what you need to know in 2017, in an easily consumable way.

"I’m not sure we learned something new from the meta-analysis, but it does give confidence that indeed there’s a good margin of safety and the risks are minimal.”