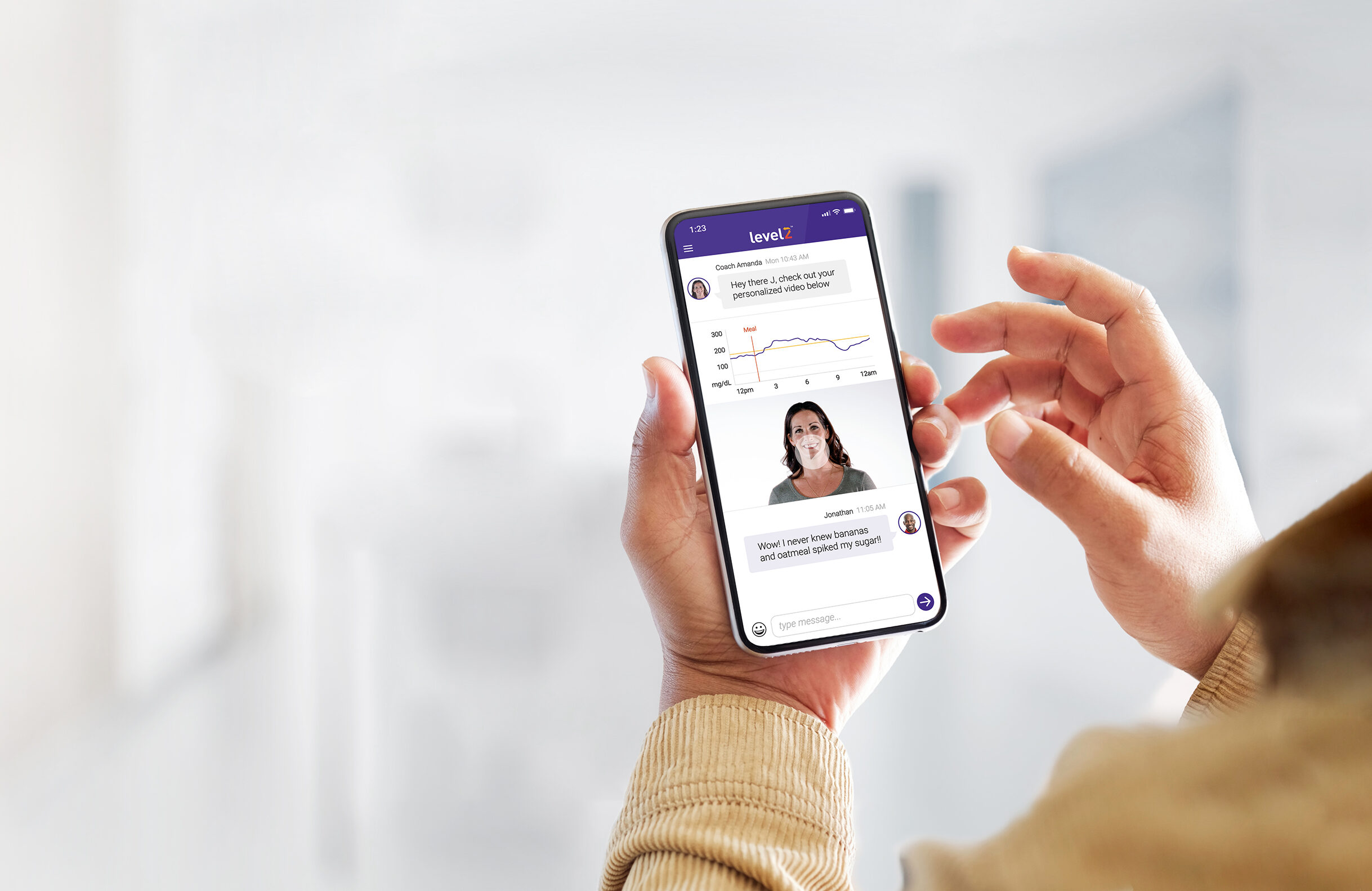
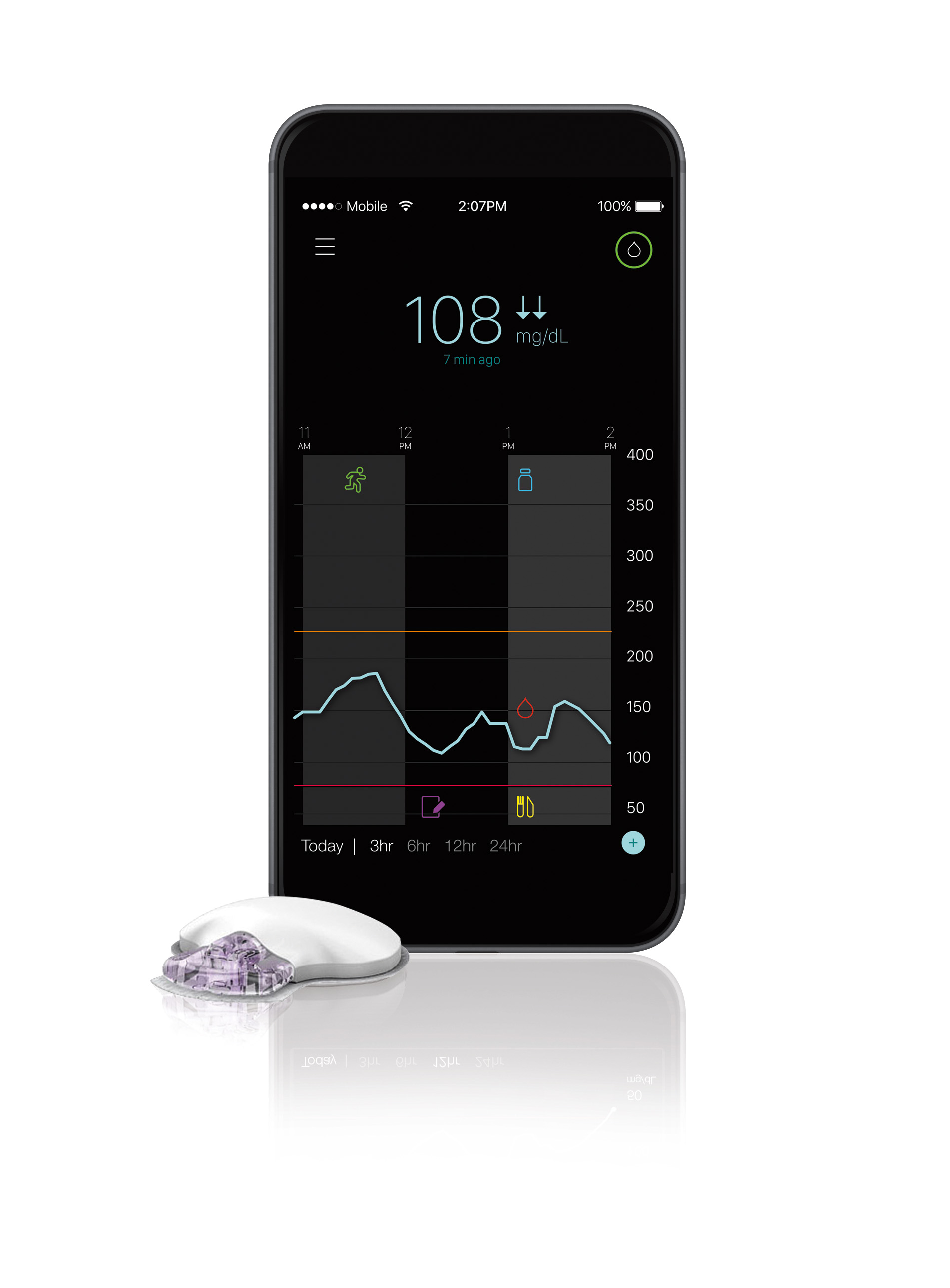
The CGM Adoption Gap Is Worse Than You Think
A new study found that continuous glucose monitors can cut healthcare costs by about 20% while also improving outcomes for insulin-dependent diabetes patients, largely by reducing hospitalizations and emergency department visits. Despite these benefits and broad insurance coverage, roughly 80% of eligible patients still are not using the technology — highlighting a significant adoption gap.