
StartUPDATES: New developments from healthcare startups
Read new developments on healthcare startups such as Seven Bridges, Scipher Medicine, Applied VR and more.

Read new developments on healthcare startups such as Seven Bridges, Scipher Medicine, Applied VR and more.

Companies developing deep brain stimulation and other neurostimulator technologies that can control and communicate signals to and from artificial body parts (i.e. limbs, eyeballs) are the latest players in the medical device sector.

Veradigm examines key clinical trends, comorbidity profiles, and treatment trends across adolescence, reproductive years, and peri-/post-menopause. Download it today!
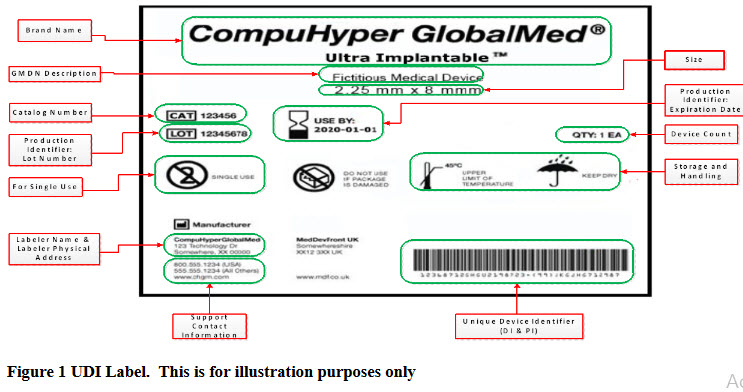
With less than 4 months for the implementation of the FDA’s third phase of the UDI system, companies with Class II devices need to ensure steps are taken to establish UDI for their products.

Health IT company RegDesk developed a network of regulatory compliance consultants across 140 countries to help life science companies navigate the path to market. Now it’s preparing to roll out a new product that would automate the process of renewing licenses for companies’ products in these countries. In a phone interview with MedCity News, the […]