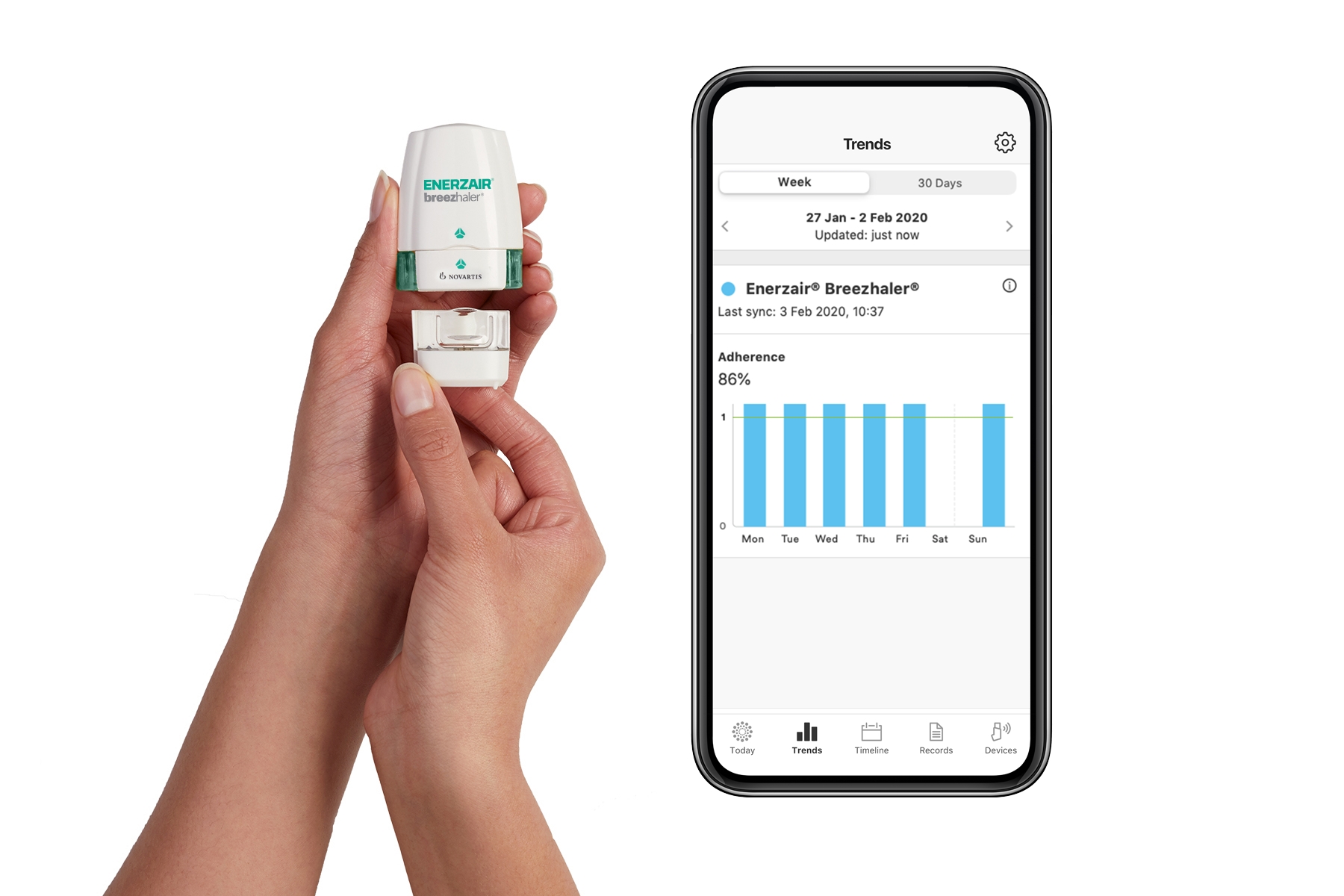
Using digital engagement to solve pain points along the patient journey: How to deliver better outcomes for patients and brands
Did you miss the event? Register for the On-Demand Webinar to learn how digital engagement can solve challenges for both patients and the brand and learn other benefits that will improve patient experience and address multiple barriers to medication adherence.