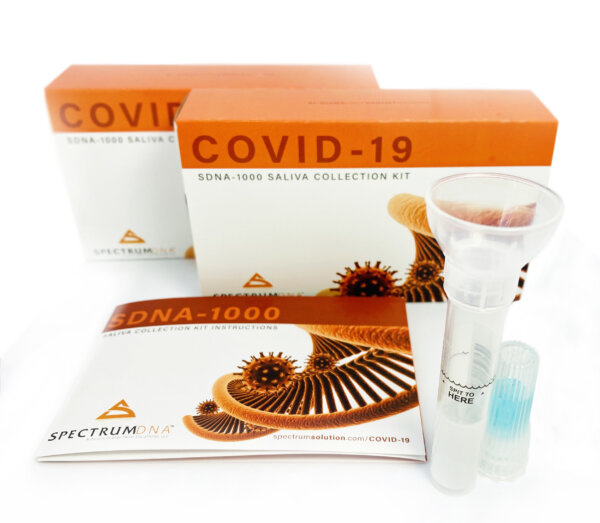
The FDA approved the first saliva -based at-home Covid-19 tests, developed by researchers at Rutgers University using Spectrum Solutions’ saliva collection device.
After the Food and Drug Administration tamped down on initial efforts to sell at-home Covid-19 test kits, startups are once again trying their hand using saliva-based tests recently authorized by the agency.
Last week, the Food and Drug Administration gave an emergency use authorization for a saliva-based Covid-19 test that can be conducted at home. The testing assay, developed by Rutgers University’s RUCDR Infinite Biologics, would make it easier for patients to swab themselves than some of the earlier Covid-19 tests, which involved swabbing deep inside the nasal canal.
In a May 8 update, the FDA specified that this is not a general authorization for at-home tests, but limited to testing performed at Rutgers Clinical Genomics Laboratory.
Just days after the approval, startups have begun to sell at-home testing kits using Rutgers’ saliva tests. Men’s health startup Vault said it will offer the tests for $150. After patients receive their test kit in the mail, they have a short Zoom call with a healthcare provider to guide them through the collection process before they ship their sample in the mail.
Mail-order wellness startup Hims and Hers plans is also offering the test for an identical price. The costs include lab fees, shipping and a telehealth consultation. Both tests will still be processed at RUCDR Infinite Biologics.
Of course, there are still some limitations to at-home testing. The FDA has said only patients with symptoms should use the saliva-based test.

A Deep-dive Into Specialty Pharma
A specialty drug is a class of prescription medications used to treat complex, chronic or rare medical conditions. Although this classification was originally intended to define the treatment of rare, also termed “orphan” diseases, affecting fewer than 200,000 people in the US, more recently, specialty drugs have emerged as the cornerstone of treatment for chronic and complex diseases such as cancer, autoimmune conditions, diabetes, hepatitis C, and HIV/AIDS.
Earlier in March, startups had moved quickly to roll out at-home test kits after the FDA approved some of the first Covid-19 tests. But the FDA quickly clarified that these tests were not approved for at-home collection, leaving many companies with a host of test kits that they couldn’t send out to patients.
Photo credit: Spectrum Solutions













