Virtual engagement platforms kept companies connected and business running in the wake of the pandemic, and they do not show signs of stopping. A recent survey published by Reuters for Within3 explores insights from over 300 medical affairs thought leaders on the role of digital work at their workplaces. A majority of those surveyed said 40-49% of medical affairs and life science engagements are expected to be virtual in the next three years, according to the Reuters survey.
Specifically, the report surveyed 304 professionals in the life science, 82% of whom held leadership positions in medical affairs, including VP or C-suite level, director, or manager positions. Of note, responses came from executives in the pharmaceutical (75%), biotech (19%), medical device (5%), and consumer health manufacturers (1%). Large companies that contributed insights to the report included Merck, Bayer, Jazz Pharma, Pfizer, and Novartis.

The Hidden Administrative Tasks Draining Small Practices
Small practices play a critical role in healthcare delivery, but they cannot continue to absorb ever-increasing administrative demands without consequences.
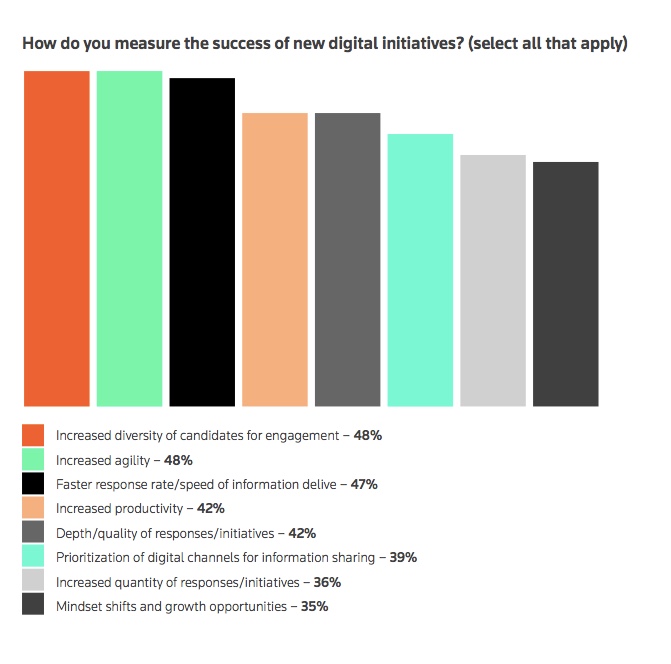
Those surveyed noted increased agility (48%), faster response rates (48%), and the prioritization of digital channels (39%) as key benefits of virtual engagement, according to the report.
“Moving into more asynchronous engagements really made us rethink the way we work and the ways we engage with the medical community,” said Ed Power, vice president of North America, medical affairs, hospital business at Pfizer in the report.
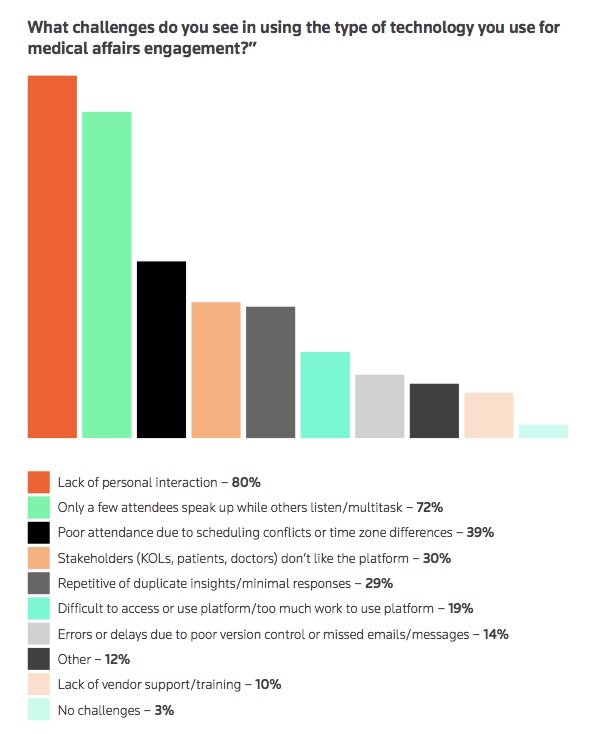
However, some external stakeholders did not like large-scale virtual medical congresses – medical professional gatherings – according to a third of respondents in the report. Despite this, 73% of respondents said stakeholders responded favorably to virtual advisory boards, while 53% approved of virtual steering committee meetings, according to the report.
“If anything, they were much richer than face-to-face meetings. People were more readily available, and often more engaged,” said Shurjeel Choudhri, SVP and head of medical and scientific affairs at Bayer about virtual advisory boards in the report. “The chat function was particularly useful in gathering nuggets of information, anecdotes and journal references. There was also greater continuity because information could be shared before and after meaning there was greater follow through.”

The Power of Real World Data to Study Women’s Health at Scale
Veradigm examines key clinical trends, comorbidity profiles, and treatment trends across adolescence, reproductive years, and peri-/post-menopause. Download it today!
Due to such perceived success of virtual options, a majority of the respondents, 65% and 85% respectively, reported they think steering committees and advisory boards will continue on as virtual events.
“For the sake of diversity, there is a strong argument for advisory boards remaining virtual with asynchronous opinion collection before and after because this uncovers previously unheard expert voices,” said Victoria Ho, director of medical excellence and capabilities at Jazz Pharma in the report.
“We’ve all been in situations – meetings or what have you – where one or two people dominate the conversation. Or where a very experienced or senior person speaks, and everyone else simply agrees because they don’t want to seem to disagree or contradict that person,” said Lance Hill, CEO of Within3. “What medical affairs and other organizations in life science have found is that a virtual environment gives everyone the opportunity to contribute their perspective – which results in much richer and diverse insights that provide more information and more direction for business strategy.”
Further, virtual events enable physicians and other HCPs to study presentations and data as their schedules allow, according to the report. Additionally, virtual events save companies time and money, the report said.
“The cost savings for us were considerable,” said Eliav Barr, SVP of global medical affairs at Merck in the report regarding the financial benefit of virtual tools to share updates quickly, from clinical trial outcomes to drug developments.
However, despite the benefits of digital collaboration, 63% of respondents noted digital fatigue as an issue, with 72% expressing frustration at other attendees multitasking during sessions or attendees not participating in discussions. Both of these reduced effectiveness of the meetings, the report said.
“Everyone is inundated with information. The market is crowded for webinars, seminars and so on. Zoom fatigue is real and we need to be aware of it,” said Kumaran Krishnan, director of medical excellence and digital transformation at Teva EU in the report.
The report indicated asynchronous virtual options for engagement as a solution to time constraints and to not prioritizing one time zone over another when scheduling. Ultimately, the bar is likely to settle at a hybrid, somewhere between virtual and in-person, the report states.
“Using virtual is giving your organization another option, another channel, for doing work. So maybe you want to have an in-person meeting, but you use an asynchronous environment to agree on your agenda ahead of time or make sure people do the pre-work – so your face-to-face time is really productive and fruitful,” Hill said regarding the mix of in-person and asynchronous work the report advocated. “Or you attend a medical conference in-person, but keep a virtual room open to collect near real-time reactions to posters or meetings with KOLs. Instead of thinking about what is lost, just acknowledge it’s a different approach, and one from which you only stand to gain.”
Photo: elenabs, Getty Images; graphs: Reuters