Alnylam Drug Approval Brings Cardiomyopathy Competition to Pfizer, BridgeBio Pharma
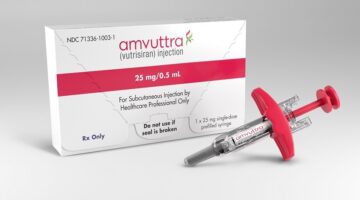
Amvuttra, an Alnylam drug approved to treat nerve pain from transthyretin amyloidosis, is now FDA-approved for treating cardiomyopathy caused by this disease. The approval brings Amvuttra to a much bigger market, but it must compete with a blockbuster Pfizer drug already established as the standard of care.