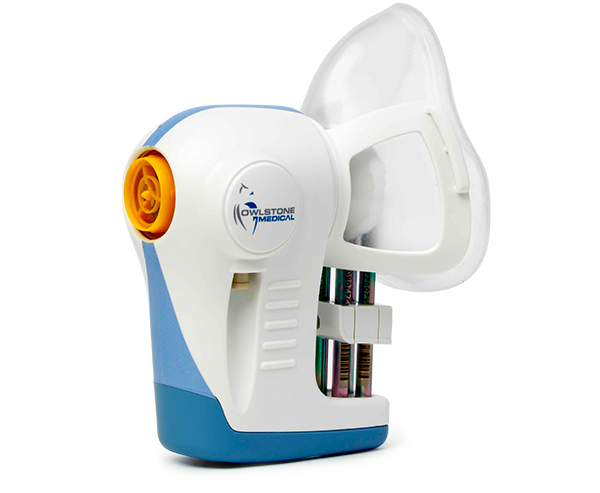
Owlstone Medical collaboration with AstraZeneca illustrates applications of breath biopsy kit
It is the latest in a series of developments for diagnostics business Owlstone Medical, which recently brought its breath biopsy kits to market.

It is the latest in a series of developments for diagnostics business Owlstone Medical, which recently brought its breath biopsy kits to market.

The move is designed to help the New Zealand device company accelerate distribution of its sensor following the opening of a U.S. office in San Mateo, California, earlier this year.

Munck Wilson Mandala Partner Greg Howison shared his perspective on some of the legal ramifications around AI, IP, connected devices and the data they generate, in response to emailed questions.

New Zealand remote monitoring company for respiratory conditions, Adherium, has opened up offices in San Mateo to pave the way for its expansion to the U.S. market.

Express Scripts members who used Propeller's inhaler tracking tools had a significant improvement in adherence to their asthma controller medication, according to the pharmacy benefit manager.

The FDA has cleared the third iteration of Adherium's adherence monitoring tool, this time for the AstraZeneca Symbicort inhaler for people with asthma and COPD.

Of the five major respiratory inhaler companies in the U.S., Propeller Health has now signed three. The latest addition, Novartis, gives Propeller an edge as it works to claim market share in the 'smart inhaler' sector projected to be worth $854 million per year by 2025.

Smart Respiratory Products is planning to make available its digital peak flow meter next year to help asthma patients to stay on top of their condition.

The deal adds spirometers and spirometry data to Propeller's platform, expanding the size of the patient population the company targets.

Rising rates of allergies worldwide will cause the global market for allergy diagnostics to nearly triple from $1.3 billion in 2015 to $3.8 billion in 2024, says a new report.
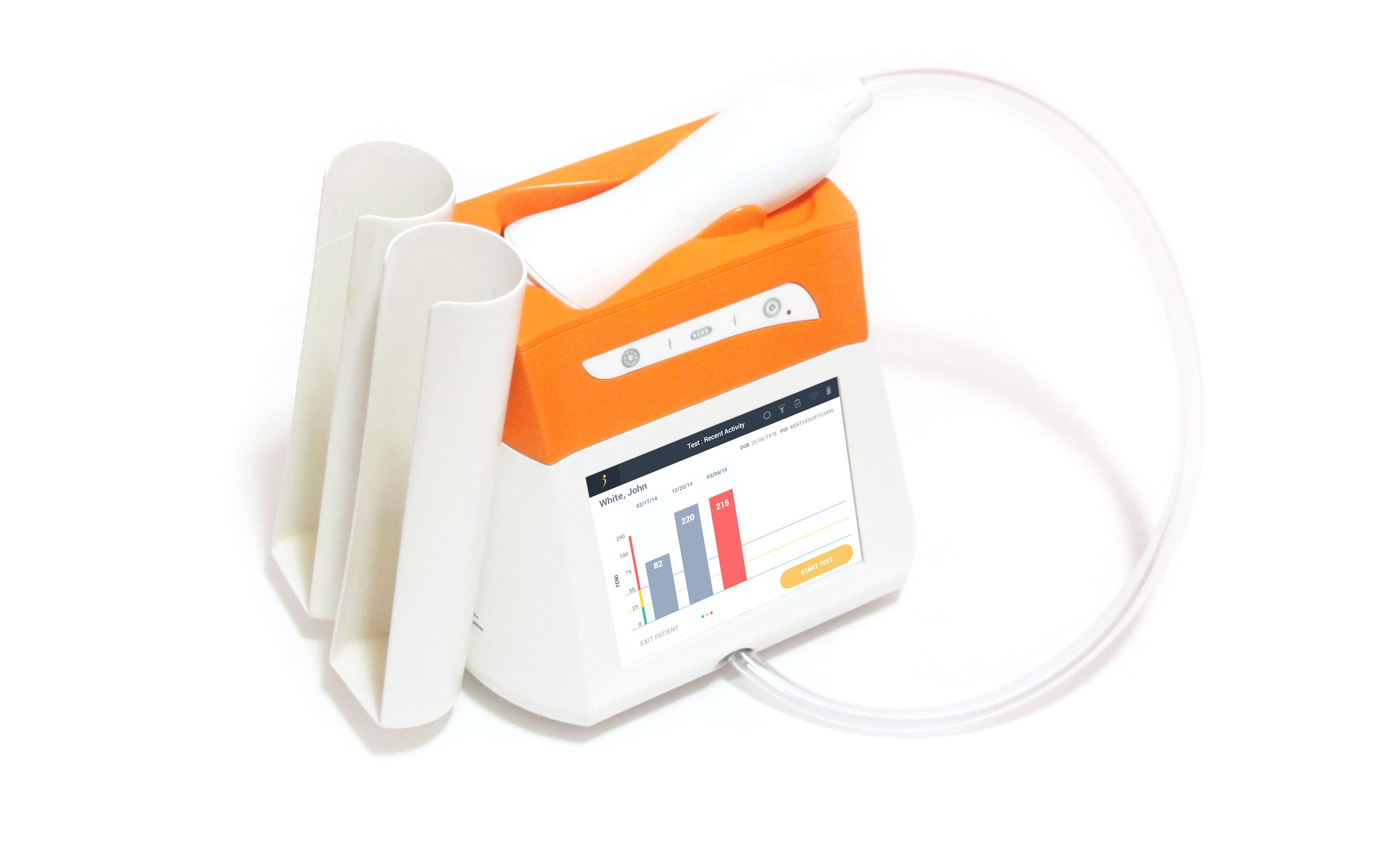
Spirometrix wants to see nearly 20 percent of pulmonologists and allergists using the Fenom Pro sensor by 2020.

We will highlight Build My Health's revenue practice management tools, which could help physician practices add up to $250,000 to their practices.
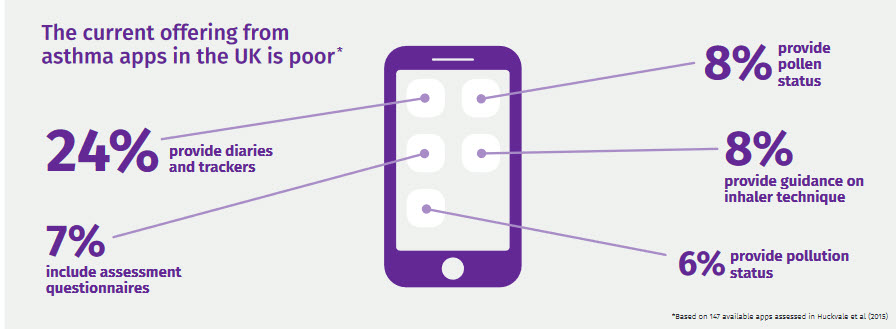
Although a report from the London-based Asthma UK supports the progress of connected devices to manage asthma, it was critical of "piecemeal" solutions that have failed to reduce complexities and guesswork from managing an episodic condition with multiple triggers.

In the next few years new products that combine drugs with digital experiences will begin to appear and will likely be more effective than regular drugs, says Chris Hogg of Propeller Health, a speaker at the upcoming MedCity ENGAGE conference in San Diego.
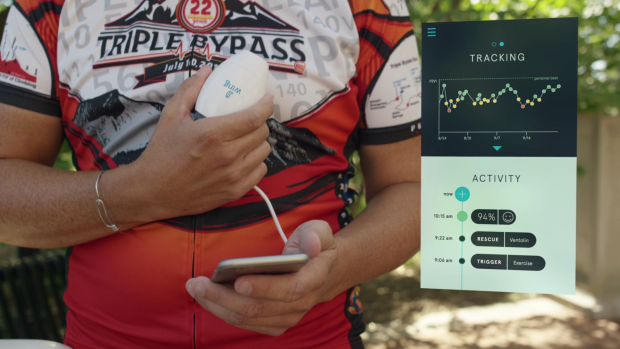
The co-founders have adopted a direct to consumer model but see interest from insurers, self-insured employers and clinical researchers.

Also, Luminex has agreed to acquire Nanosphere and Boston Children's Hospital has published a guide on Bring Your Own Devices in hospital settings.

A recent study at Dignity Health revealed that patients who used the Propeller Health platform saw greater improvement in symptom- free days than patients receiving routine care. And adults who experienced uncontrolled asthma also saw improvements.