
As Propeller Health closes latest fundraise, it expands digital drug delivery ambitions beyond inhalers
The goal is to expand its connected device capabilities beyond asthma and COPD inhalers to include other diseases and drug delivery devices.

The goal is to expand its connected device capabilities beyond asthma and COPD inhalers to include other diseases and drug delivery devices.
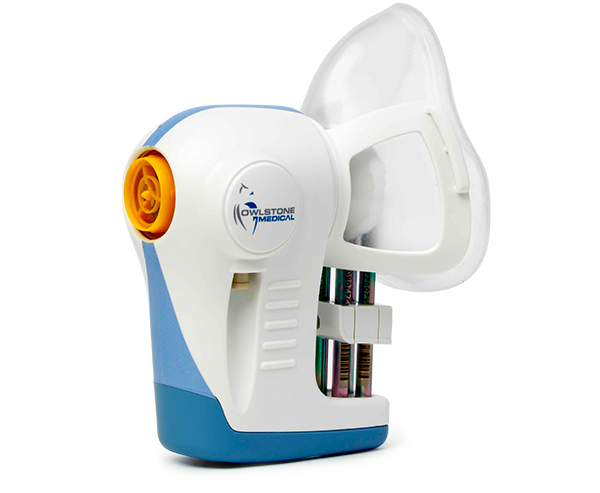
It is the latest in a series of developments for diagnostics business Owlstone Medical, which recently brought its breath biopsy kits to market.

Munck Wilson Mandala Partner Greg Howison shared his perspective on some of the legal ramifications around AI, IP, connected devices and the data they generate, in response to emailed questions.

The move is designed to help the New Zealand device company accelerate distribution of its sensor following the opening of a U.S. office in San Mateo, California, earlier this year.

New Zealand remote monitoring company for respiratory conditions, Adherium, has opened up offices in San Mateo to pave the way for its expansion to the U.S. market.

The study will seek to determine whether Owlstone Medical's breath biospy tool can match the right patient for the right treatment and to assess the treatment effects of a novel drug for COPD.

Express Scripts members who used Propeller's inhaler tracking tools had a significant improvement in adherence to their asthma controller medication, according to the pharmacy benefit manager.

The FDA has cleared the third iteration of Adherium's adherence monitoring tool, this time for the AstraZeneca Symbicort inhaler for people with asthma and COPD.
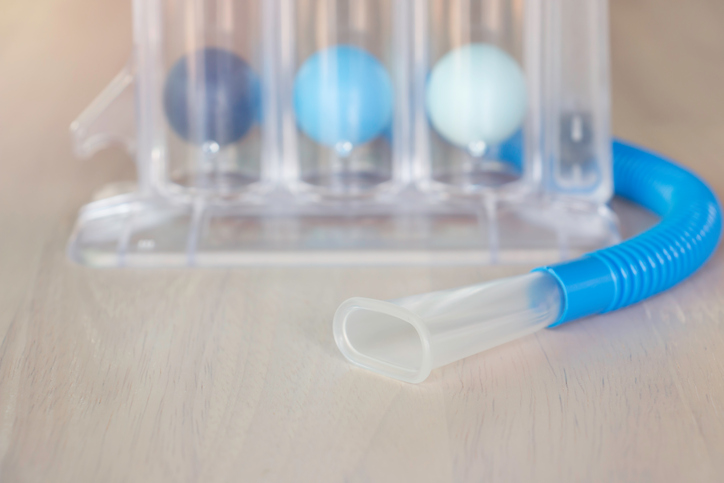
Cost-effective, easy-to-use tools, like spirometers need to be implemented as the standard similar to a blood pressure test in doctor’s offices and other locations where people receive their care.

Of the five major respiratory inhaler companies in the U.S., Propeller Health has now signed three. The latest addition, Novartis, gives Propeller an edge as it works to claim market share in the 'smart inhaler' sector projected to be worth $854 million per year by 2025.

In the next few years new products that combine drugs with digital experiences will begin to appear and will likely be more effective than regular drugs, says Chris Hogg of Propeller Health, a speaker at the upcoming MedCity ENGAGE conference in San Diego.

Gabby Everett, the site director for BioLabs Pegasus Park, offered a tour of the space and shared some examples of why early-stage life science companies should choose North Texas.

Patients on Boehringer Ingelheim drugs for respiratory conditions have the option to enroll with certain health systems to get free access to inhalers embedded with Propeller Health sensors.

The move is part of a broader trend of major respiratory brands to develop connected versions of their medications with sensors integrated with inhalers.
Enabling a constructive patient/physician dialogue means that physicians need to invite patients to become equal partners in their own care, and to give voice to every patient’s uniqueness.

Investors included Blue Cross Blue Shield of Massachusetts, Bioadvance, Three Leaf Ventures — an affiliate of the Broe Group, Swanhill Capital and StartUp Health through its digital health syndicate.

Pond Healthcare Innovation CEO Daniel Taub said in an interview that the Air Smart spirometer device is CE-certified by European regulators and is available in Europe