Medtronic Exec: AI Is Improving Device Launches & Patient Outcomes
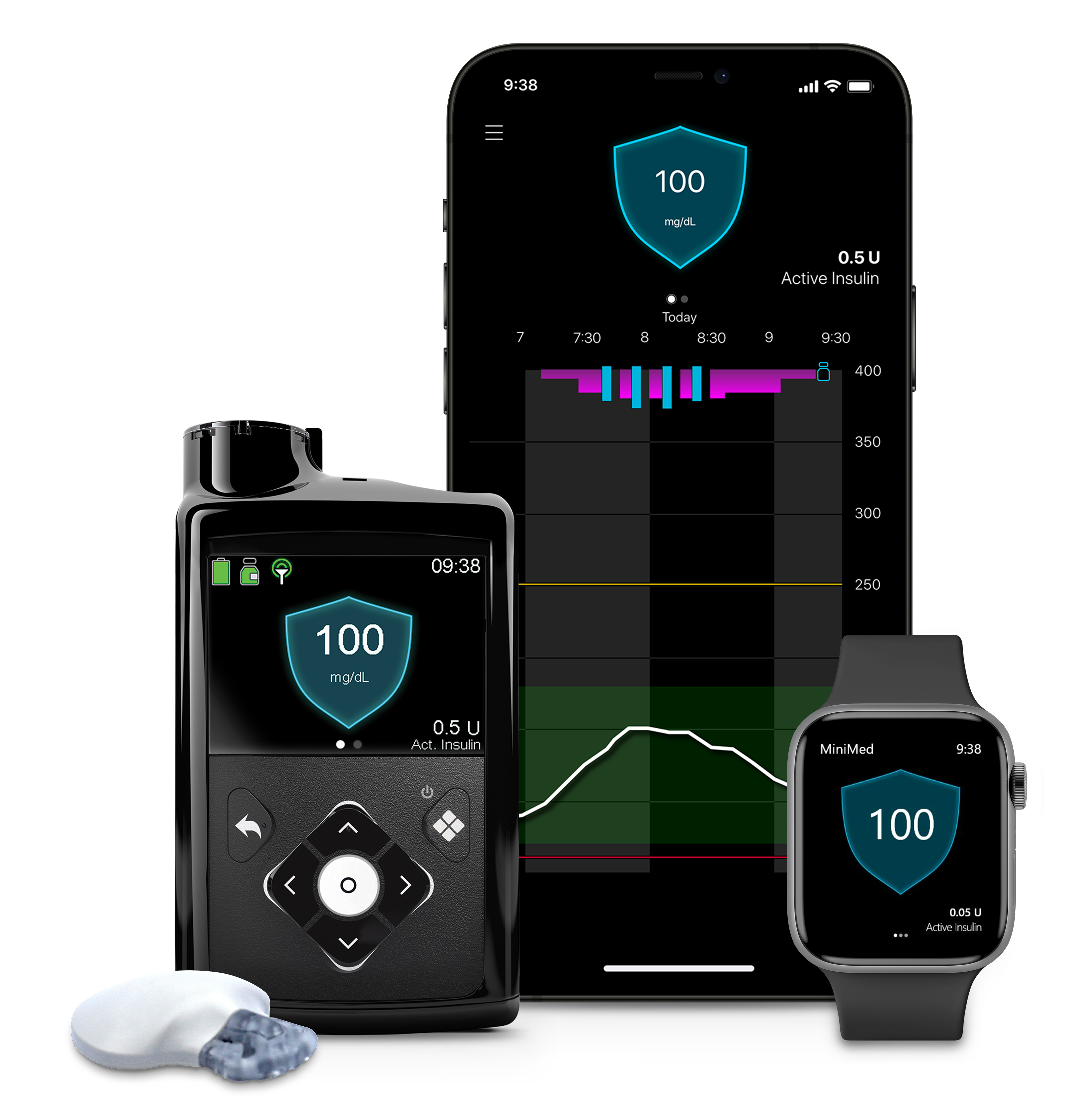
Jennifer Thompson, Medtronic’s vice president of U.S. aortic sales, detailed how AI is transforming medical device commercialization during a fireside chat at Reuters’ MedTech conference. The technology is helping teams identify patients, optimize their sales deployment and discover care bottlenecks.