
Here’s an example of product adoption with zero reimbursement
At a recent medical device conference in San Francisco, a Medtronic executive gave an example of a device finding adoption even though the device is not reimbursed.

At a recent medical device conference in San Francisco, a Medtronic executive gave an example of a device finding adoption even though the device is not reimbursed.

Medical device startups have a huge opportunity in developing devices to treat ischemic and even hemorrhagic stroke and stand to win reimbursement with good clinical and economic evidence.

This eBook, in collaboration with Care Logistics, details how hospitals and health systems can facilitate more effective decision-making by operationalizing elevated awareness.

Medtronic becomes the first in the U.S. to win FDA approval for a quadripolar cardiac resynchronization therapy pacemaker that is also safe to use under MRI if certain conditions are met.

Medtronic is rumored to be selling its medical supplies business to Cardinal Health in shift to become a disease management company eager to play a role in the continuum of care as opposed to a widget maker.

Cardiac mapping has been an important tool to make ablations safe and effective, but it has required catheterization, unlike Medtronic's new product.

Medtronic CEO Omar Ishrak joins the board of Patient Safety Movement Foundation founded by Masimo CEO Joe Kiani and takes the pledge to share data, something Kiani had wanted for years.

In an era of escalating healthcare costs and a growing preference for natural, holistic approaches to health, The Impact Brands emerges as a collective of diverse brands dedicated to supporting overall wellness through natural means.

Partnerships between insurers, health systems and medtech companies as well as insight gleaned from Big Data are going to key to the success of value-based care in the medical devices industry.
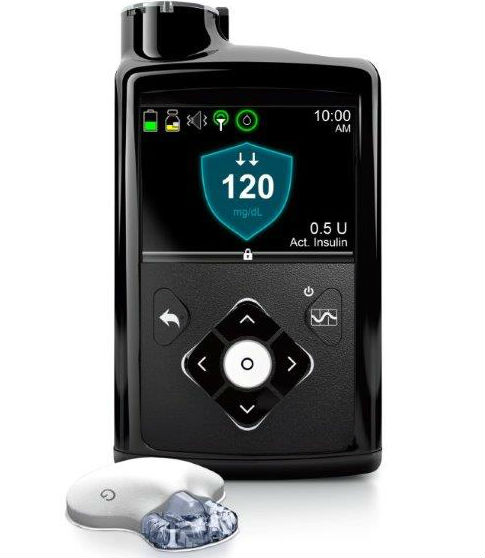
FDA approves the first automated insulin delivery system giving Medtronic the distinction of being the first in a category that is set to revolutionize daily disease management for type 1 diabetes patients.

Medtronic launched a next-gen insulin pump in mid August, but analysts are more interested in when the FDA will approve the company's breakthrough device - the artificial pancreas.
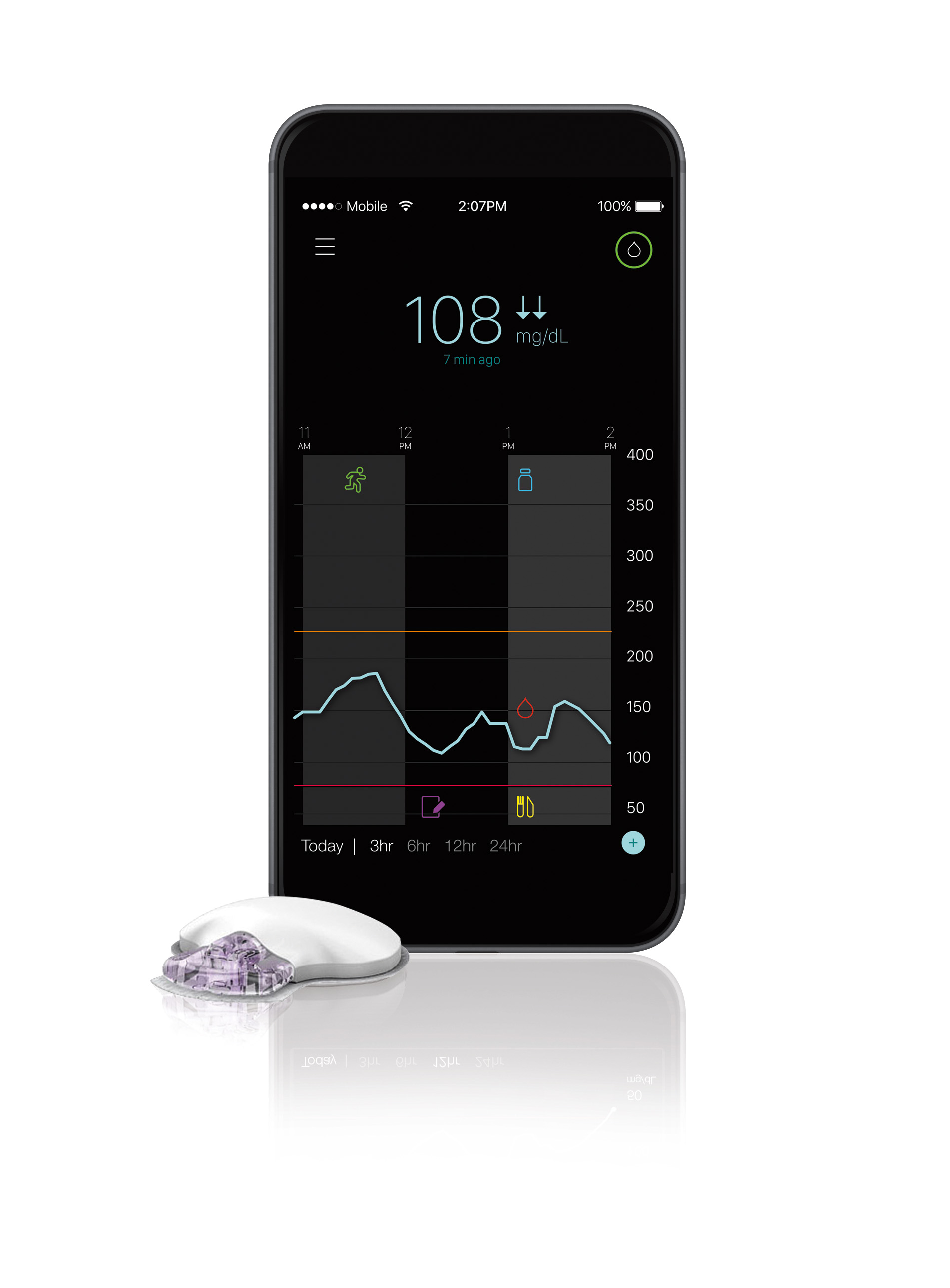
Medtronic is giving diabetes patients on insulin injection therapy outside the U.S. the ability to view blood glucose data from their continuous glucose monitors on their smartphones.

Canada has a proud history of achievement in the areas of science and technology, and the field of biomanufacturing and life sciences is no exception.

Also, Owlstone has spun out Owlstone Medical to develop a breathalyzer for clinical diagnostics to battle cancer and Pocket DNA sequencers will become ubiquitous, according to Dr. Eric Topol.

Medtronic wants to serve heart failure patients with moderate or severe symptoms with a mix of devices and services.

Also, out-of-pocket costs per hospitalization soar, AstraZeneca seeks to extend Crestor patent under the Orphan Drug Act and Virginia Mason's CEO decides to stay after all.

Medtronic will also work with Canary Health to develop programs that enlist Medtronic’s devices, services and infrastructure and Canary Health’s behavior change programs, design, and user engagement insights.

A low-cost, connected continuous glucose monitor that can provide clinical decision support seems like it would be a nice addition to the toolset of Diabeter, a Danish operator of Diabetes clinics, which Medtronic acquired last year.