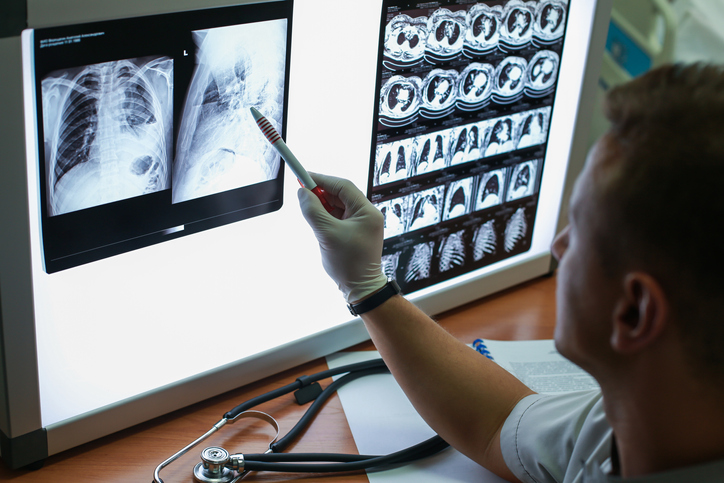
RapidAI gets FDA clearance for AI designed to detect pulmonary embolism
The company's AI-enabled imaging software identifies a suspected pulmonary embolism and delivers real-time notifications to physicians so the patient can be treated quickly.

The company's AI-enabled imaging software identifies a suspected pulmonary embolism and delivers real-time notifications to physicians so the patient can be treated quickly.

The regulatory clearance would allow AIdoc’s system to be used to detect incidental pulmonary embolism, meaning that it could flag unsuspected findings in CT scans.

The FDA decision, which builds on a prior approval of the company's algorithm for the detection of intracranial hemorrhages through CT scans, is part of a larger vision sketched out by CEO Elad Walach of plugging new algorithms into the Aidoc system to create a new standard of care in radiology.

Texas startup BiO2 raised $4 million for its catheter that prevents pulmonary embolisms in patients prone to get them - particularly those with severe head and spinal injuries.