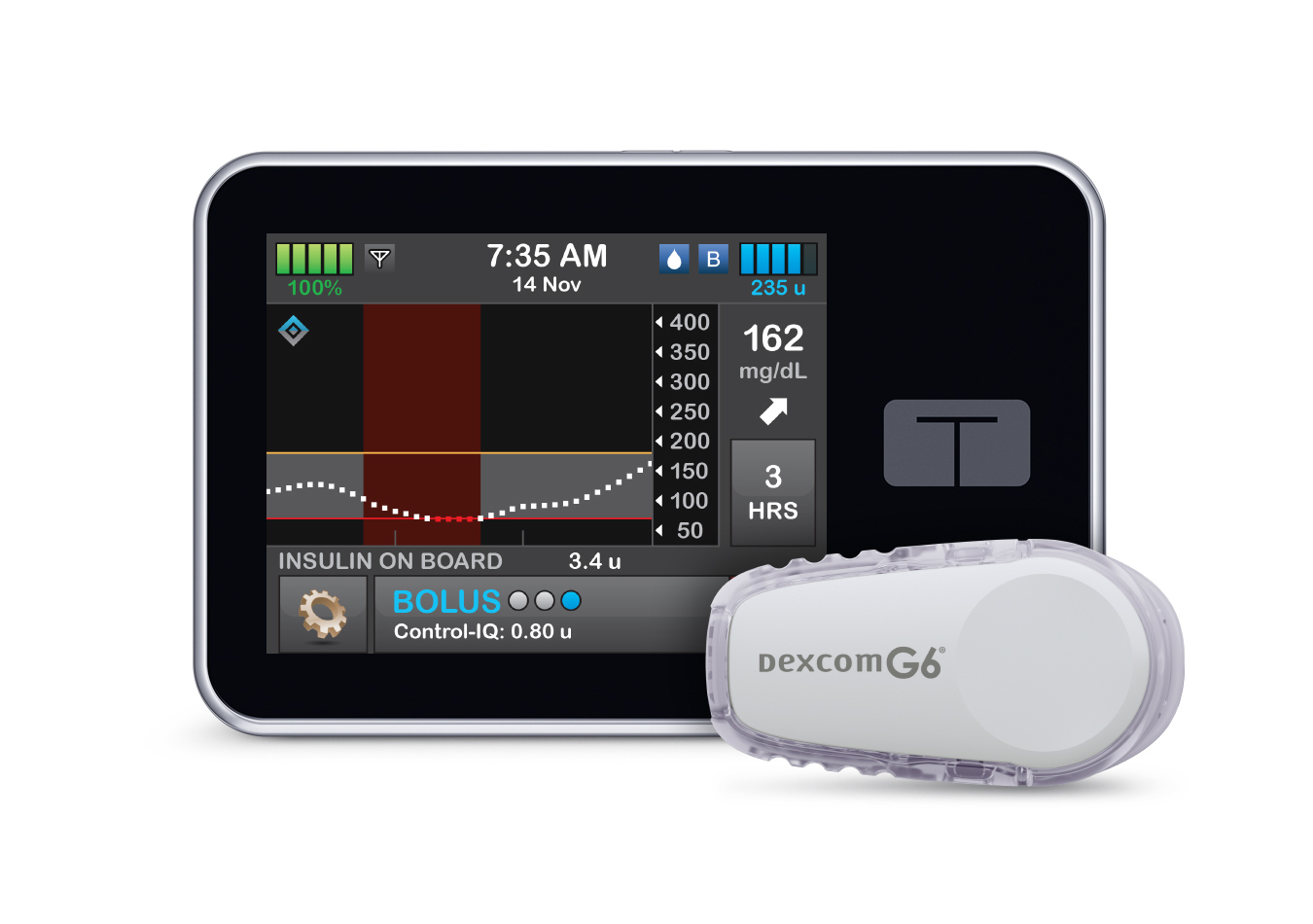
The Surprising Rivalries-Turned-Friendships That Are Shaping Diabetes Technology
Over the past decade, Abbott and Dexcom — the two largest makers of continuous glucose monitors — have been teaming up with medical device companies that sell automated insulin delivery systems. Industry experts think these partnerships are smart moves to stay relevant in the rapidly-developing diabetes care space.