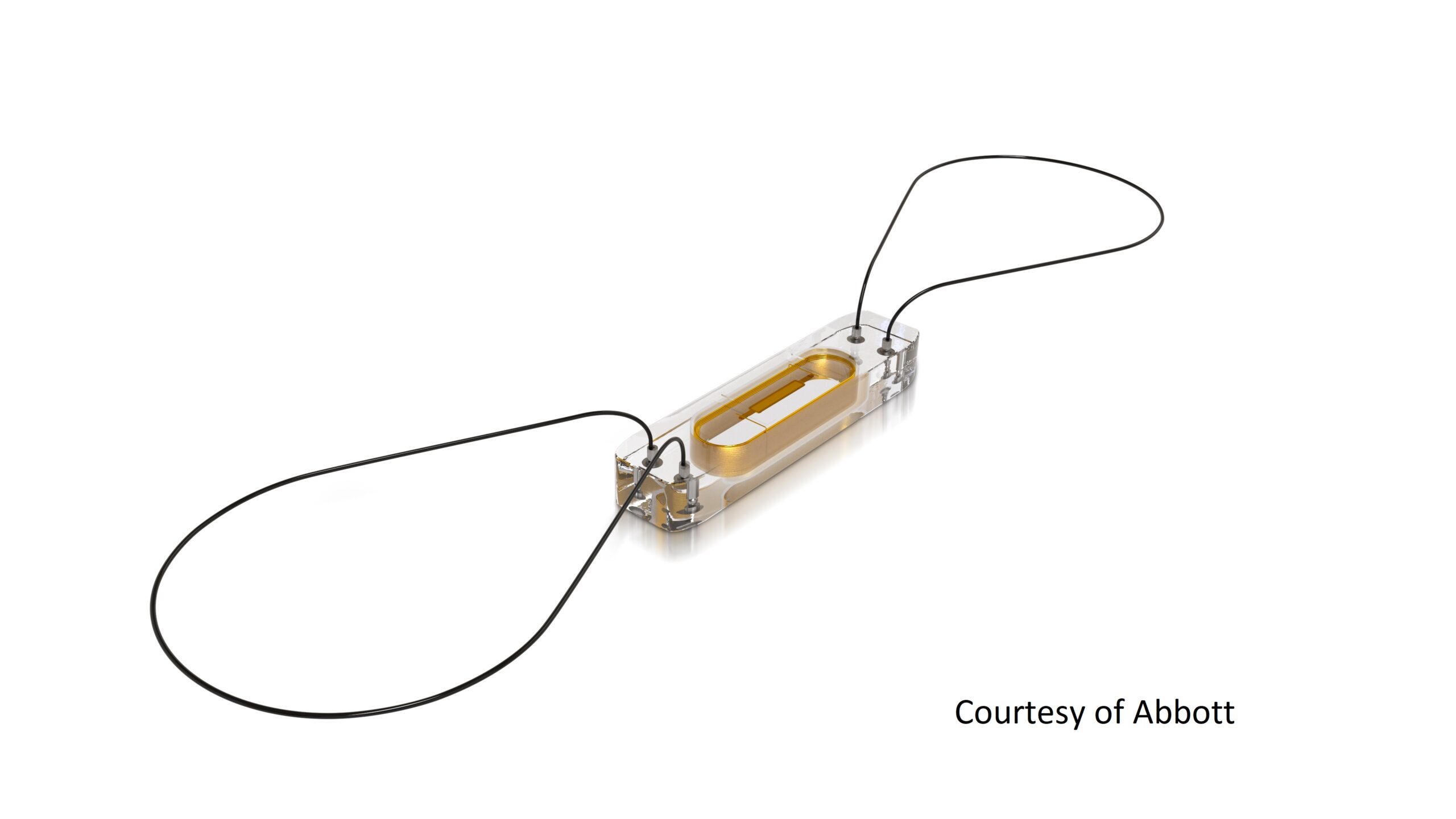
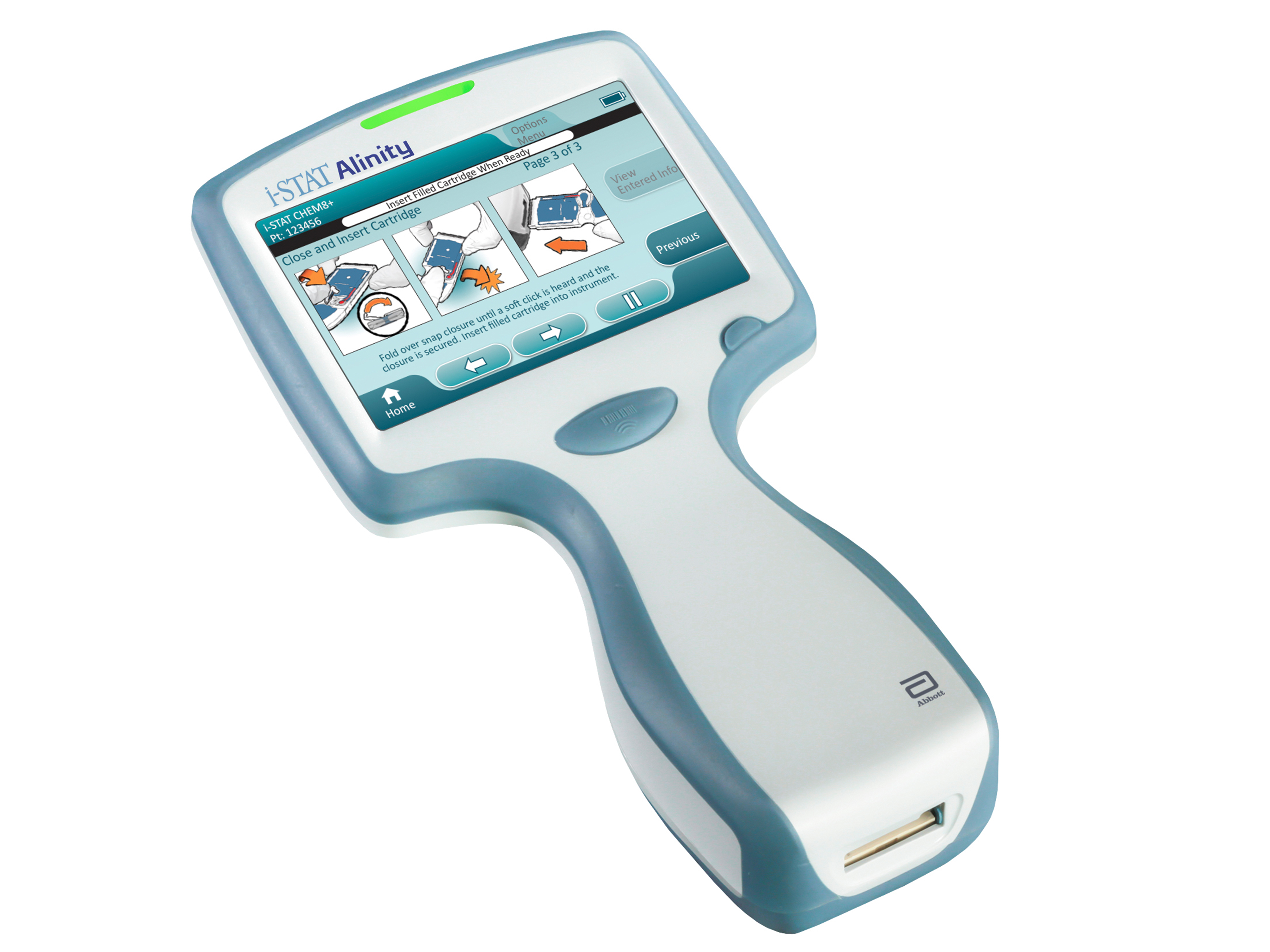
Medical device companies should consider embracing connected care platforms
A recent webinar hosted by MedCity News and sponsored by BioT shared insights on some of the ingredients to successful creation and execution of remote patient monitoring solutions supported by a cloud-based connected care platform with speakers from BioT and Abbott.