Eisai Alzheimer’s Drug Gets FDA Decision Date; Advisory Meeting Is Planned
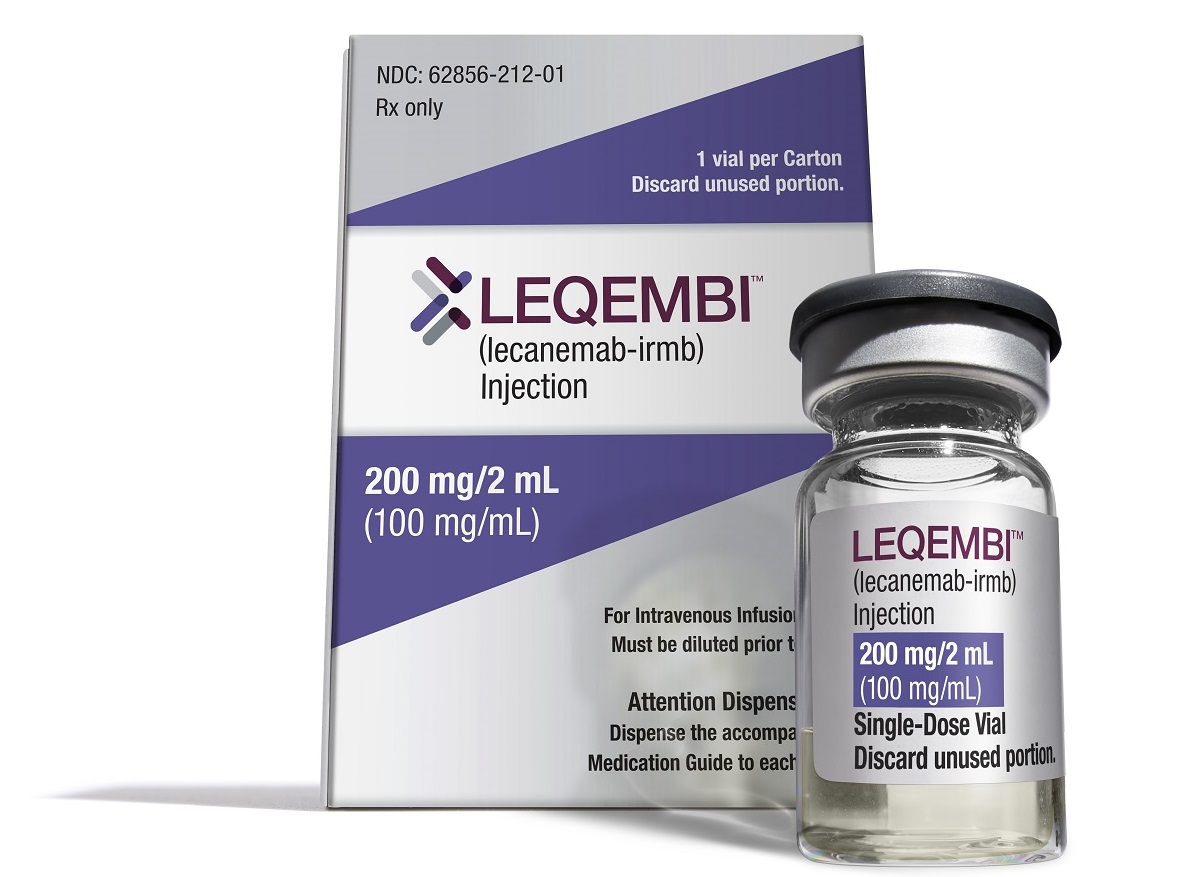
Leqembi, the Eisai Alzheimer’s disease drug awarded accelerated FDA approval in January, could receive a decision on full approval in early July. But in setting the target date for a regulatory decision, the agency also said it plans to convene an advisory committee meeting to discuss the drug.