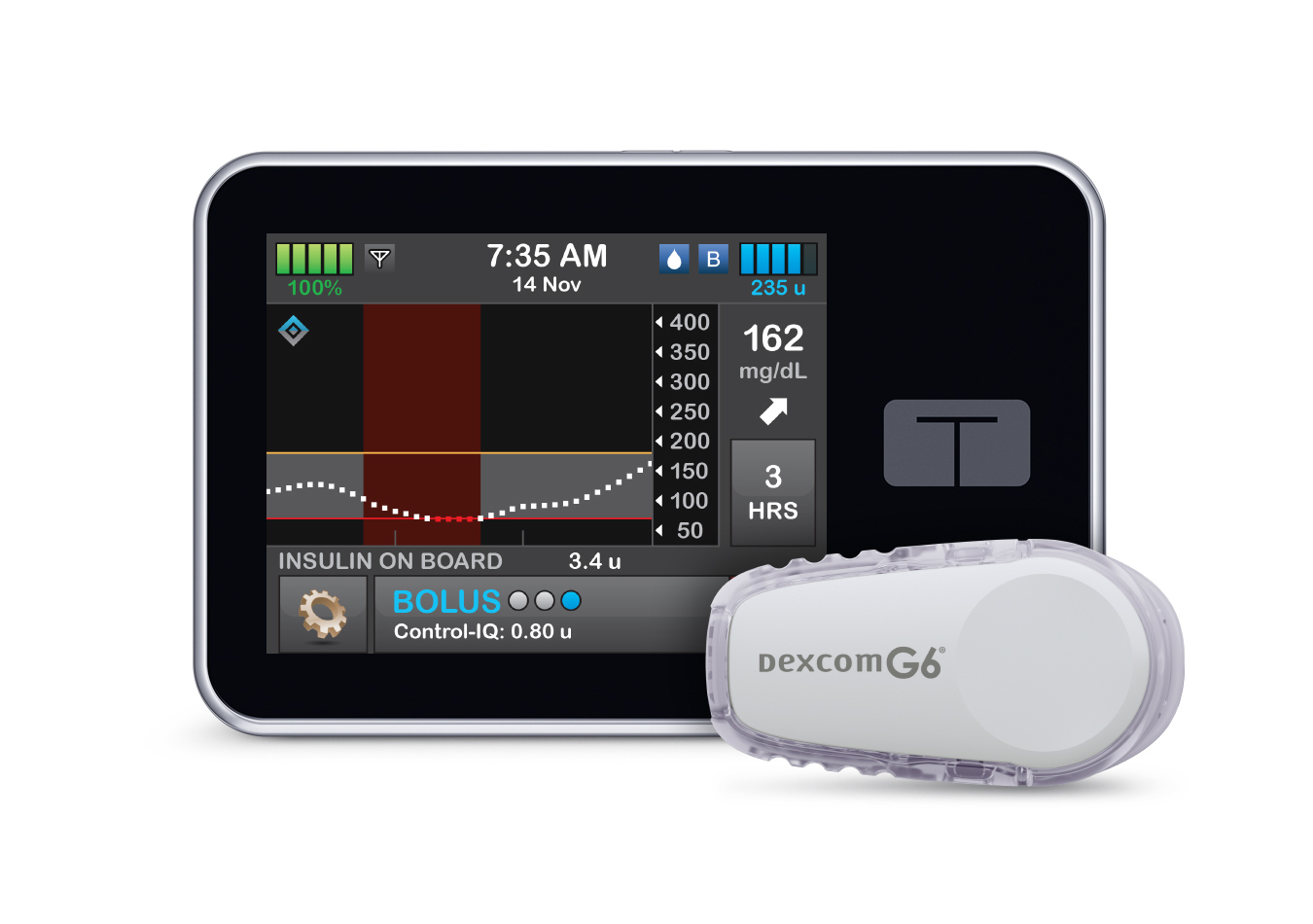
Tandem Diabetes snags FDA approval for interoperable insulin pump software
Tandem Diabetes’ technology will create a new approval pathway for predictive dosing software that can be used with other devices.

Tandem Diabetes’ technology will create a new approval pathway for predictive dosing software that can be used with other devices.

Abbott struck a deal with Bigfoot Medical by which the latter will use Abbott's glucose sensing technology to develop an artificial pancreas and not that of DexCom, the CGM market leader.

Munck Wilson Mandala Partner Greg Howison shared his perspective on some of the legal ramifications around AI, IP, connected devices and the data they generate, in response to emailed questions.
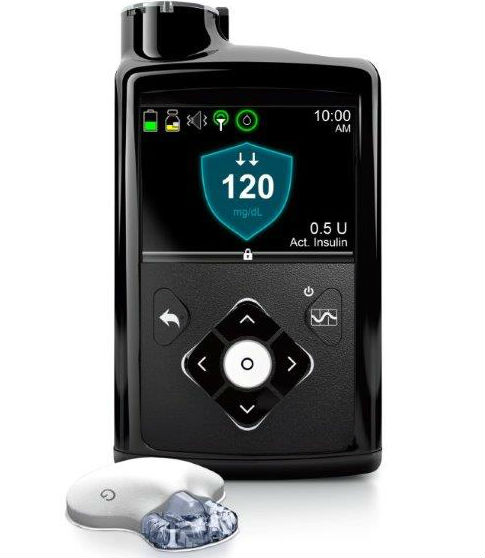
FDA approves the first automated insulin delivery system giving Medtronic the distinction of being the first in a category that is set to revolutionize daily disease management for type 1 diabetes patients.
The tubeless insulin pump maker enrolls first patients in feasibility study to tests it artificial pancreas technology.

Bigfoot Biomedical is building an artificial pancreas, combining continuous glucose monitor tech with an insulin pump - and raising some capital to do so.

A 4-year-old boy in Australia, Xavier Hames, just received the first artificial pancreas device from Princess Margaret Hospital for Children, which will regulate his insulin levels for him. The hospital was where clinical trials for the device have been taking place for several years, and now it’s available for $8,100. This is big news for the many […]

San Diego’s ViaCyte is clearly up to plenty – the regenerative medicine company just announced it has teamed up with Janssen Research & Development, securing $20 million in financing in the process. As part of the deal, the pharma giant gains rights to evaluate clinical efficacy of ViaCyte’s Type 1 diabetes treatment – an artificial […]