
Moderna does one better than Pfizer/BioNTech; touts 94.5% efficacy for its Covid vaccine
The company is the second this month to report promising results for a Covid-19 vaccine even as the virus rages across the U.S.

The company is the second this month to report promising results for a Covid-19 vaccine even as the virus rages across the U.S.
Interferons serve as the body’s first line of defense against infection, sounding the alarm and activating an army of virus-fighting genes. Lab studies show interferons are suppressed in some people with Covid-19, perhaps by the virus itself.

Munck Wilson Mandala Partner Greg Howison shared his perspective on some of the legal ramifications around AI, IP, connected devices and the data they generate, in response to emailed questions.
When all key health stakeholders are connected at the hub, common insights can be leveraged, allowing all healthcare providers to respond intelligently, without delay and ultimately make the best decisions possible for their patients.

Fear and uncertainty regarding the coronavirus have made online groups targets for the spread of false information and to help fellow patients, some of these groups are making it a mission to stamp out misinformation.
News of positive results from Pfizer's Covid-19 vaccine is heartening but historically, important scientific announcements about vaccines are made through peer-reviewed medical research papers that have undergone extensive scrutiny about study design, results and assumptions, not through company press releases.

The news sent the stock market soaring as it represents a key milestone in developing and distributing a vaccine for the virus. Still public confidence in vaccines need to be boosted, an expert noted.

This eBook, in collaboration with Care Logistics, details how hospitals and health systems can facilitate more effective decision-making by operationalizing elevated awareness.

Many of the strategies that were on the bottom of the priority list in early 2020 may have moved up, with a new focus on digital enablement and other technological capabilities.
CMS' interim rule states that Medicare will cover Covid-19 vaccines approved by the FDA, including those receiving emergency use authorization, in a reversal from its usual policy. The vaccine will be made available at no cost to Medicare beneficiaries.

Deferred care because of Covid-19 will lead to additional stress on the healthcare system in the very near future and here's how insurers can manage it.

Even though Covid-19 is distinct from any health crisis the country has previously faced, assessing U.S. public health strategies during past crises can provide insight into what has contributed to a more or less equitable response.

There is a surging demand for pre-sterilized surgical technologies at healthcare facilities because of the desire to both curb risk factors associated with Covid-19 at hospitals and also reduce the possibility of bacterial co-infections.

To keep critical revenue flowing, hospitals and ambulatory surgery centers need to resume elective surgeries and procedures as quickly as possible and mobile apps can help by giving patients and staff vital information, provide advance symptom screening and help providers reduce elective procedure no-shows through better patient compliance.

The Food and Drug Administration approved Gilead's antiviral drug as the first treatment for Covid-19 but just last week a WHO report showed that the drug appeared to have little to no effect on hospitalized patients.

While there are encouraging signs of reimbursement falling in step with the move towards a more value-based healthcare system, what is needed now to further encourage healthcare innovators is to properly rationalize approval processes imposed by the FDA and CMS.
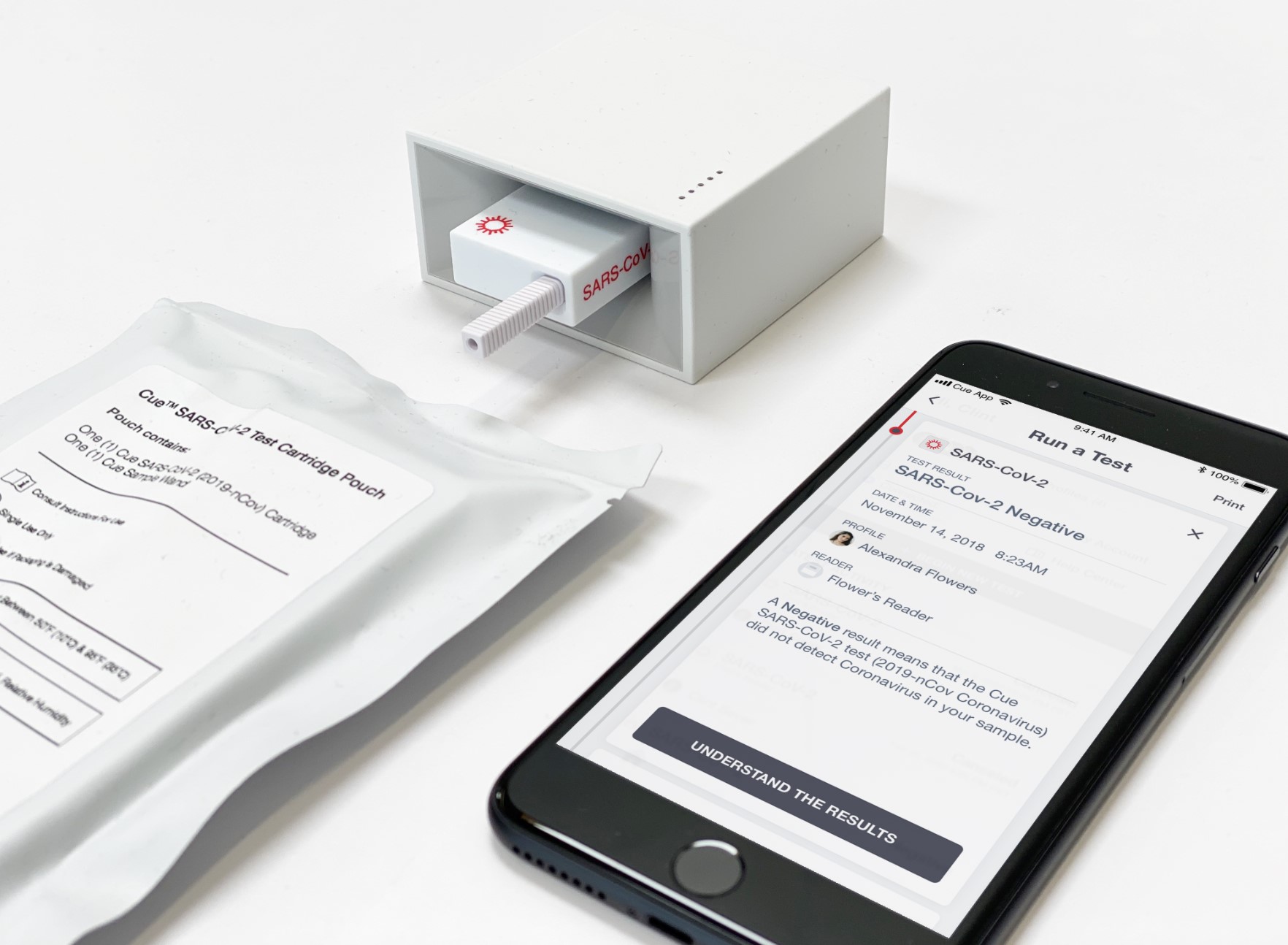
San Diego-based diagnostics startup Cue Health won a $481 million from the Department of Health and Human Services and the Department of Defense to produce more coronavirus tests. The company received an emergency use authorization in June for its rapid molecular test.