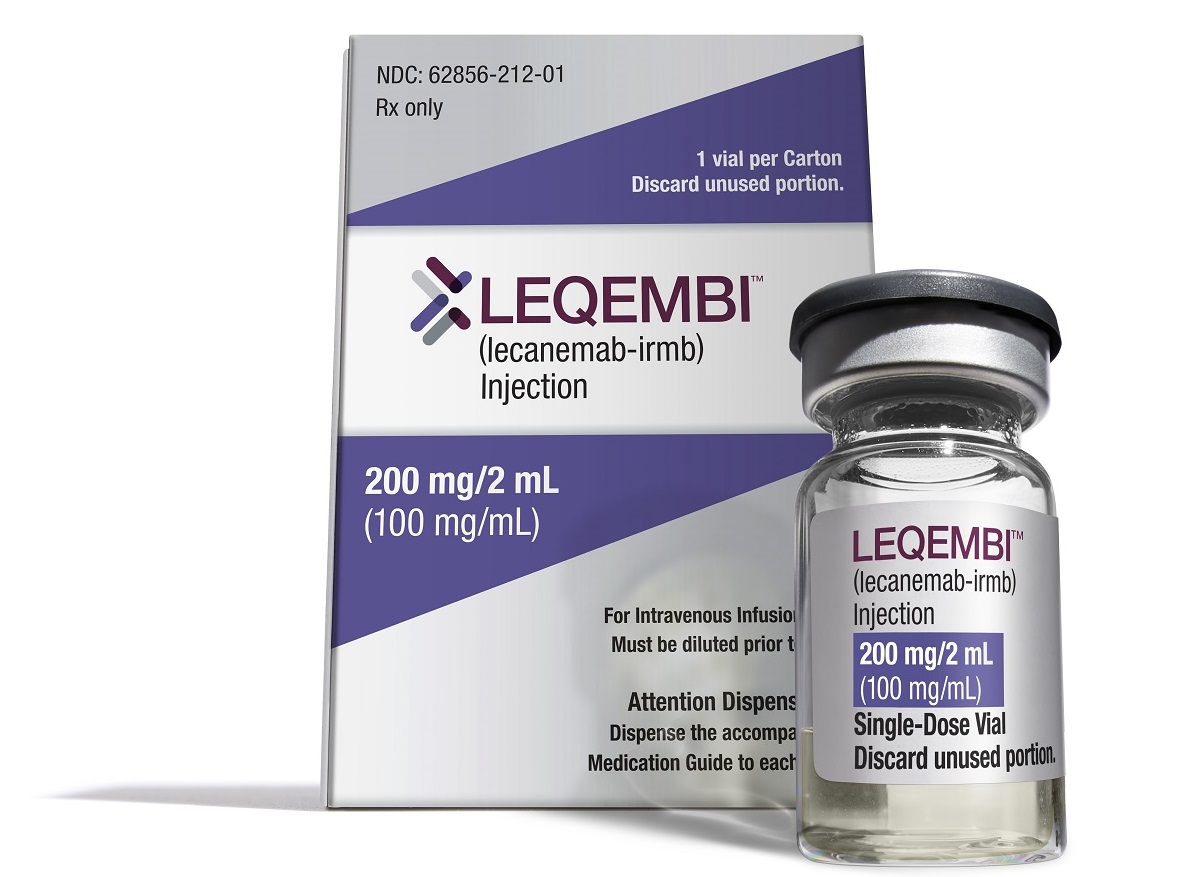
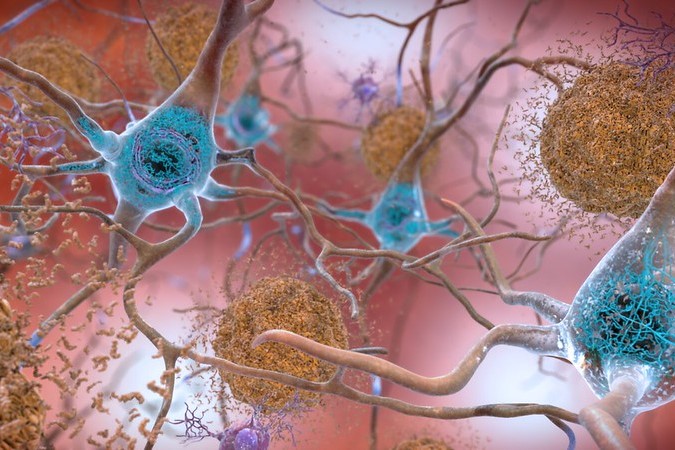
Alzheimer’s Therapy Is Coming to the Home With FDA Approval of Injectable Eisai, Biogen Drug
Injectable Leqembi permits weekly maintenance dosing of the Eisai and Biogen Alzheimer’s disease drug that may be done at a patient’s home. Other recent notable regulatory news includes several rare disease drug approvals and narrower FDA nods for Covid-19 vaccines.