
FDA approves Novo Nordisk’s oral Type 2 diabetes drug
The drug, Rybelsus, is the first oral medication in the GLP-1 receptor agonist class. The approval was based on data from 10 trials that enrolled more than 9,500 patients.

The drug, Rybelsus, is the first oral medication in the GLP-1 receptor agonist class. The approval was based on data from 10 trials that enrolled more than 9,500 patients.

An analyst called the data, on ZGN-1061, encouraging in a relatively small study but added that more preclinical data will be needed to give a clearer picture of the way forward for the drug.

Munck Wilson Mandala Partner Greg Howison shared his perspective on some of the legal ramifications around AI, IP, connected devices and the data they generate, in response to emailed questions.
An analyst wrote that with selonsertib's failures in fourth- and now third-stage fibrosis, the drug will have no place in NASH treatment. Gilead has entered partnerships focused on other compounds for the fatty liver disease.
The deal comes days after Gilead partnered on clinical development in NASH with Denmark's Novo Nordisk, and two months after the high-profile failure of a Phase III study in the disease.

PatientPoint will give patients and physicians AMA-developed educational material to help them identify the risks for the diseases and encourage them to take action.

The company, one of several developing drugs for the growing fatty liver disease, said in an SEC filing that it would sell its shares at $26.33 apiece.

Prior to founding health benefits platform Jiff (which was acquired by Castlight in 2017), Newell worked for years in the remote care monitoring and chronic disease management space.

In contrast to 23andMe's previous FDA approvals for genetic risk tests for conditions like Alzheimer's, the diabetes assessment did not go through the FDA clearance pathway because it was characterized as a wellness product not intended to make diagnoses and provide medical advice.

During the three-month program, UMHP members with type 2 diabetes will use emocha's app to submit daily videos of themselves taking their medication.

With investments in big data and cloud computing, physicians and public health experts are collaborating with engineers to design frameworks capable of performing large-scale population health research, including in diabetes.

We will highlight Build My Health's revenue practice management tools, which could help physician practices add up to $250,000 to their practices.
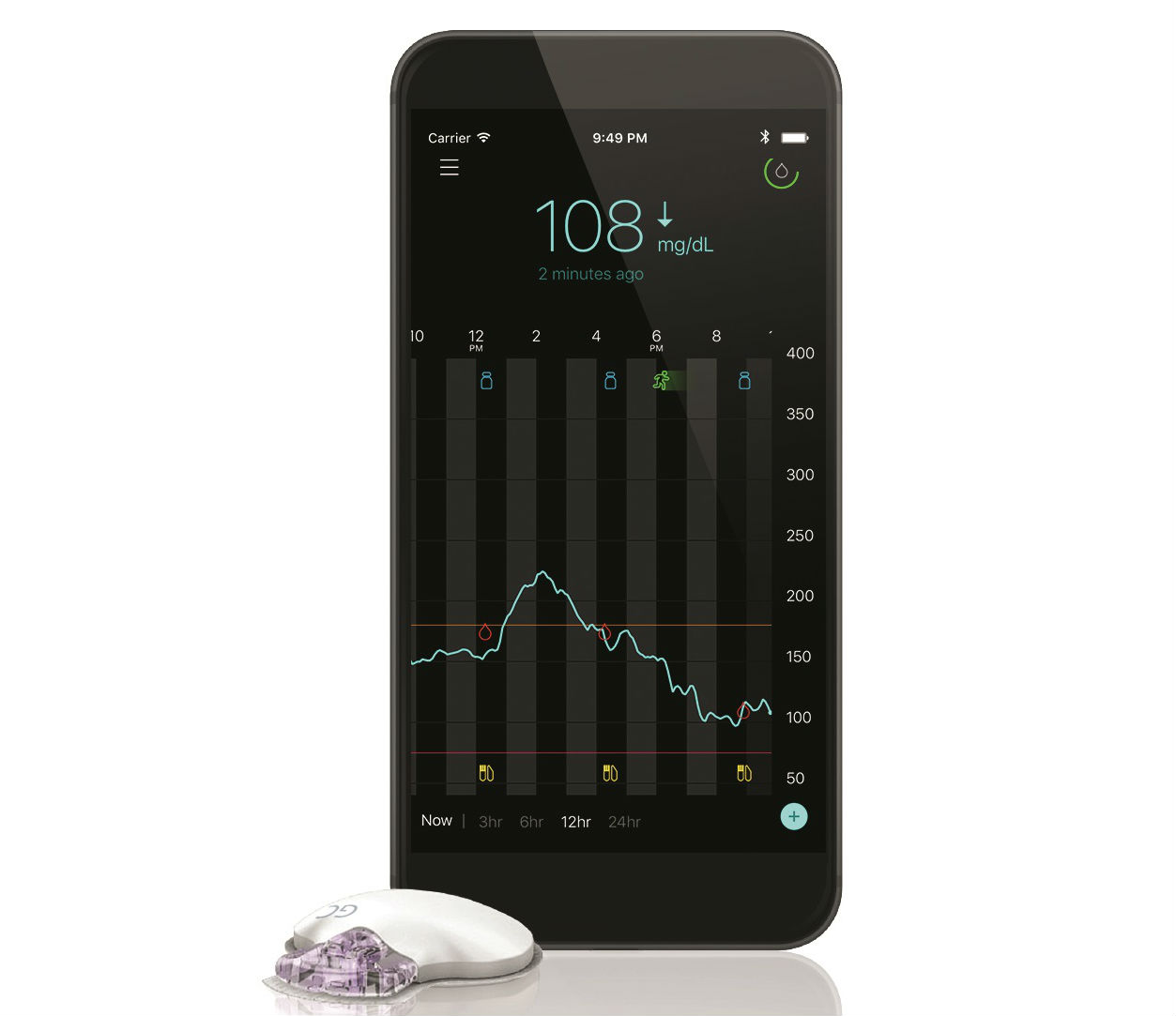
Medtronic had previously betted on higher adoption of its insulin pumps tethering the use of continuous glucose monitors to those pumps but has recently returned to the standalone CGM market with the launch of its Guardian Connect device in the U.S. Should Dexcom and Abbott worry?
In a bid to expand into the larger, more lucrative type 2 diabetes market, the medical device maker offers a performance guarantee to payers and employers tied to its MiniMed 670G insulin pump system.

Russ Johannesson pushed back on the idea that he is the CEO whose sole goal is to take the company to an exit, opined on competitors like Livongo, and said Glooko is the connective tissue across the diabetes management ecosystem.

With the FDA approval of a new continuous glucose monitoring system with machine learning capabilities to predict highs and lows in blood-glucose levels, Medtronic makes a bigger play for type 2 diabetes patients.

Fractyl Laboratories has raised a $44 million Series D financing round to build upon a chance discovery that bariatric surgery can resolve Type 2 diabetes in a subset of patients — independent of any weight loss.