INVEST Conference Video: The future of at-home testing beyond Covid-19
Covid-19 has renewed interest in diagnostics and a way to deliver care at home. Learn from companies and experts as they address the future of these two developments.

Covid-19 has renewed interest in diagnostics and a way to deliver care at home. Learn from companies and experts as they address the future of these two developments.

After insurers were mandated to cover at-home Covid-19 tests, more people tested at-home screenings for other conditions, speakers said in a discussion hosted by MedCity INVEST. As preventive tests declined significantly during the pandemic, insurers also turned to creative means to give people access.

Munck Wilson Mandala Partner Greg Howison shared his perspective on some of the legal ramifications around AI, IP, connected devices and the data they generate, in response to emailed questions.

After the Food and Drug Administration authorized a swathe of over-the-counter tests for Covid-19, pharmacies are preparing to stock their shelves. But at $20 to $100 a pop, will enough people use them to make a difference?

After a busy year, at-home testing company Everlywell is growing through two acquisitions. It bought telehealth practice PWNHealth and another at-home lab testing company.
As Covid-19 joins the ranks of cancer and chronic diseases to be tracked and prevented through testing, health plans will need testing solutions that can be distributed quickly and at scale.

After Everlywell received an emergency use authorization last year from the FDA for its at-home collection kit, the agency gave Everlywell the green light to sell its test kits without requiring a prescription.

Ellume, whose at-home Covid-19 test was cleared by the FDA in December, got a $232 million contract from the Department of Health and Human Services to deliver 8.5 million tests.

Few people question whether care delivery will continue to migrate out of the hospital into new settings such as the home. The better question is, what services - specifically - will migrate to the home, and what innovations are necessary to support these migrations?

During a panel discussion at the J.P. Morgan Healthcare Conference, three physicians from Stanford Medicine discussed the investment opportunities that can help accelerate innovation in healthcare in the coming year.

The Austin-based startup has distributed more than 500,000 Covid-19 test kits to date, and expects total sales to quadruple by the end of the year.

Gabby Everett, the site director for BioLabs Pegasus Park, offered a tour of the space and shared some examples of why early-stage life science companies should choose North Texas.

The retailer is offering at-home Covid-19 testing kits, including nasal swabs and saliva test kits, from Los Angeles-based startup myLab Box.

Humana will send 1 million screening kits to its members for managing diabetes and screening for colorectal cancer. The at-home tests are intended to offset screenings and other preventive care being pushed back due to Covid-19.
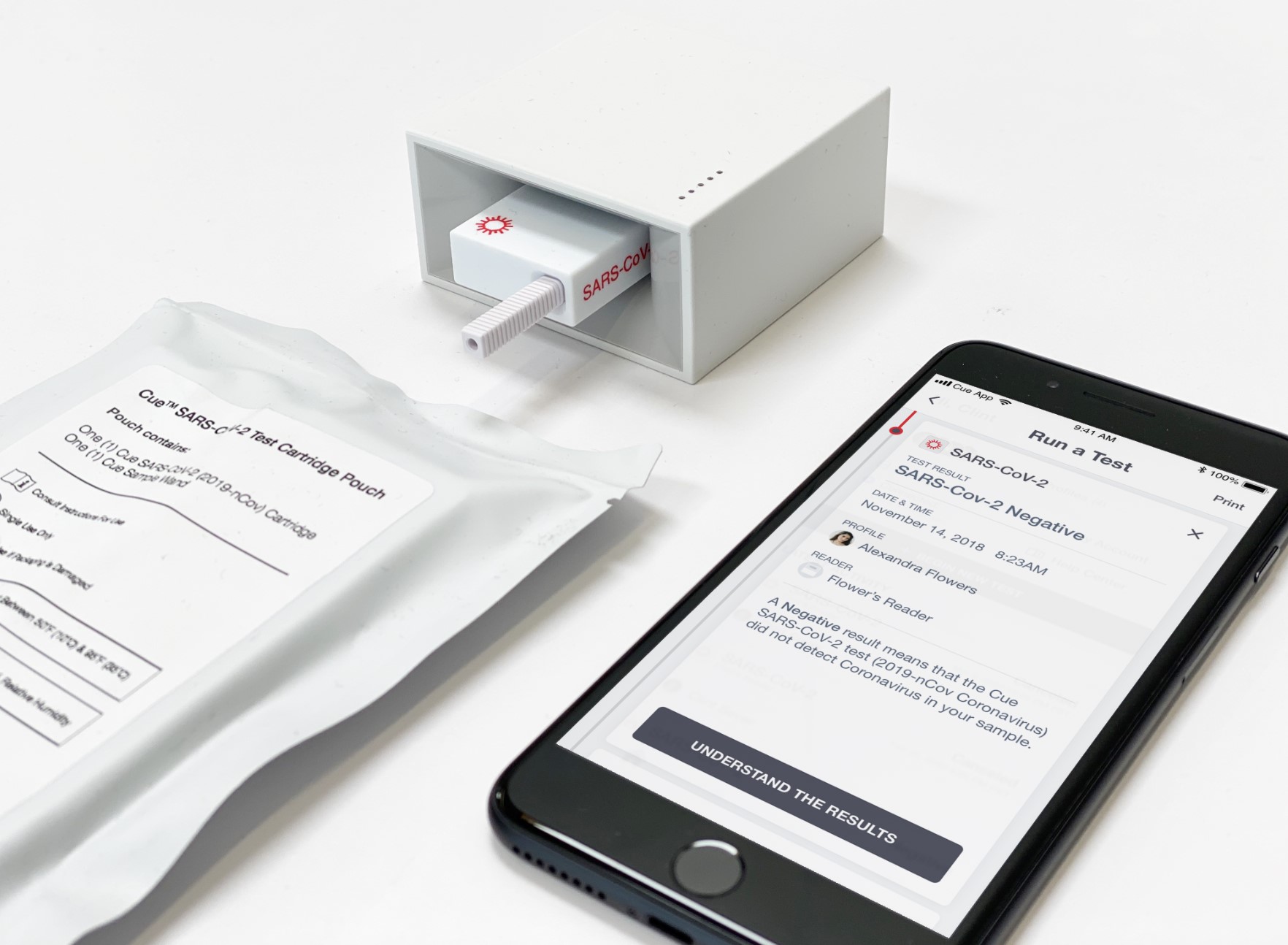
Cue Health, a startup developing a connected testing device, closed a $100 million series C round. The company recently submitted its system to the FDA for emergency use authorization to be used for onsite Covid-19 testing.

GRAIL, a startup developing cancer liquid biopsy tests, raised a $390 million series D round. Read more about which companies raised funding this week.
New guidance from the FDA this week would make it easier for patients to self-test for Covid-19. But the agency still hasn’t approved home testing.